Volume 11, Number 11—November 2005
Dispatch
Methicillin-resistant Staphylococcus aureus in Taiwan
Abstract
We found a virulent closely related clone (Panton-Valentine leukocidin–positive, SCCmec V:ST59) of methicillin-resistant Staphylococcus aureus in inpatients and outpatients in Taiwan. The isolates were found mostly in wounds but were also detected in blood, ear, respiratory, and other specimens; all were susceptible to ciprofloxacin, gentamicin, and trimethoprim-sulfamethoxazole.
Although most methicillin-resistant Staphylococcus aureus (MRSA) illness and death are associated with healthcare facilities (H-MRSA), isolates from community-associated MRSA (C-MRSA) infections have been obtained with increasing frequency in the last few years in different countries, including Taiwan (1–5). The changing epidemiology of MRSA has become an important public health concern worldwide (1,4). MRSA arises when S. aureus organisms acquire a large mobile genetic element called staphylococcal cassette chromosome mec (SCCmec) (6). Most H-MRSA strains possess either SCCmec II or III; most C-MRSA strains possess SCCmec IV (7–9). Recently, a novel type V SCCmec type was characterized and found in a C-MRSA isolate from Australia (10). With the exception of variable resistance to erythromycin, C-MRSA strains are generally susceptible to other non-β-lactam antimicrobial agents, in contrast to most H-MRSA, which are typically resistant to many of the non-β-lactam agents (1,4,9). Another characteristic of C-MRSA is the production of Panton-Valentine leukocidin (PVL), an extracelluar cytotoxin involved in primary skin infections and pneumonia (2–4).
We conducted a study to characterize the molecular epidemiology of selected MRSA isolates from the Taiwan Surveillance of Antimicrobial Resistance (TSAR), a national surveillance program of inpatient and outpatient clinical isolates in Taiwan (11). We describe the finding of a virulent closely related clone of MRSA and its prevalence in Taiwan.
A total of 398 and 865 nonduplicate S. aureus isolates were collected from March to May 2000 from 21 hospitals and from July to September 2002 from 26 hospitals, respectively, as part of the TSAR collection (11). The proportions of isolates for the 2 years were similar for outpatients (27.5% in 2000 and 28.9% in 2002); the rest of the isolates were from inpatients. The most common specimen type was wound, which accounted for 35.4% and 49.2% of all S. aureus isolates in 2000 and 2002, respectively. Antimicrobial susceptibility was determined on the basis of results of MICs obtained from a broth microdilution method, following the guidelines of the National Committee for Clinical Laboratory Standards (12) by using custom-designed Sensititre plates (Trek Diagnostics, East Essex, United Kingdom). Overall, MRSA accounted for 238 (59.8%) of 398 S. aureus isolates in 2000 and 475 (54.9%) of 865 S. aureus isolates in 2002.
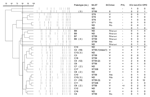
To obtain an overall understanding of MRSA throughout Taiwan, we first chose 80 MRSA isolates (68 inpatient and 12 outpatient isolates) collected in 2002 from 4 hospitals located in the north, middle, south, and east regions of Taiwan. Pulsed-field gel electrophoresis was performed according to a published protocol (9), and pulsotypes were assigned to clusters of isolates with >80% similarity from the dendrograms. SCCmec typing and PVL gene detection were performed according to published protocols (7,8,10). Multilocus sequence typing (MLST) was performed on randomly selected strains from major pulsotypes, and the sequence type (ST) was assigned by using the MLST database (http://www.mlst.net) (13). Three major clusters (pulsotypes) were found, including 47 (58.8%) pulsotype A, 7 (8.8%) pulsotype B, and 18 (22.5%) pulsotype C (Figure A1). All 47 pulsotype A isolates had SCCmec III; 4 isolates tested by MLST had ST239. In addition to being resistant to clindamycin (94%), erythromycin (100%), and tetracycline (100%), all pulsotype A isolates were resistant to ciprofloxacin (CIP), gentamicin (GEN), and trimethoprim/sulfamethoxazole (SXT). The 7 pulsotype B isolates possessed SCCmec IV but were not of the 4 known IV subtypes (IV not a–d); all were CIP- and SXT-susceptible but GEN-resistant. Seventeen of the 18 isolates in pulsotype C possessed SCCmec V; the other had SCCmec IVa; all 18 were CIP/GEN/SXT-susceptible. Of the 10 isolates tested by MLST from pulsotype B (3 isolates) and pulsotype C (7 isolates), all had ST59. Only pulsotype C isolates were PVL-positive.
Because we found a large percentage (21.3%, 17/80) of SCCmec V:ST59, PVL-positive clones, we selected an additional 69 CIP/GEN/SXT-susceptible MRSA isolates (25 from 2000, 44 from 2002) to determine what portion of them had the same clone. These isolates were from intensive care unit (ICU) and non-ICU inpatients, plus outpatients from 15 hospitals in 2000 and 21 hospitals in 2002 (excluding the 4 hospitals already tested). Fifteen (60%) of the 25 isolates from 2000 and 32 (72.7%) of the 44 isolates from 2002 belonged to pulsotype C, had SCCmec V, and were PVL-positive. Of the 4 isolates characterized by MLST, 2 were ST59, 1 was ST388, and 1 was a new ST; the last 2 isolates differed from ST59 by 1 nt each in gmk and arcC genes, respectively.
When the 2 groups of isolates were combined, a total of 90 CIP/GEN/SXT-susceptible MRSA were studied (Figure A2). These isolates were mostly resistant to clindamycin (89%), erythromycin (92%), and tetracycline (82%). Of these 90 isolates, 68 (75.6%) were PVL-positive and belonged to same pulsotype C; 64 (71.1%) had SCCmec V. MLST performed on 12 isolates found 10 to be ST59; the other 2 (ST338 and the new ST) are closely related to ST59. Forty-eight of these 64 isolates were from wounds (75%), but isolates were also found in respiratory (6 isolates), ear (3 isolates), blood (2 isolates), urine (2 isolates), and catheter and other body site (3 isolates) specimens. Only half (32 isolates) were from outpatients; the rest were from ICU (10 isolates) and non-ICU (22 isolates) inpatients. Two ICU isolates were from hospital-acquired infections. Because 189 (26.5%) of the 713 MRSA isolates from the 2000 and 2002 collections were CIP/GEN/SXT-susceptible, and because 64 (71.1%) of the 90 CIP/GEN/SXT-susceptible MRSA we studied were PVL-positive, SCCmec V:ST59, and belonged to pulsotype C, an estimated 18.8% (26.5% × 0.711 = 18.8%) of MRSA isolates in Taiwan could be this closely related virulent clone.
The reason for the high prevalence of this virulent clone (pulsotype C:ST59, SCCmec V, PVL-positive) of MRSA in Taiwan in both inpatients and outpatients is not known. Data on the prevalence of SCCmec V are still limited, and ST59 has been described infrequently. A recent longitudinal study of MRSA isolates in the San Francisco area found that the ST59-SCCmec IV has increased steadily from 1999 to become 1 of the 4 major clones associated with C-MRSA (14).
Production of the PVL cytotoxin is considered a genetic marker for C-MRSA, and although PVL-positive MRSA have usually been associated with skin and soft tissue infections, severe and fatal infections, such as necrotizing pneumonia, have been reported (2–4). Thus, PVL may confer an additional virulence advantage for this particular clone of MRSA in Taiwan. Other possible explanations are that the less resistant C-MRSA can grow faster than multidrug-resistant H-MRSA and that SCCmec IV and V carried by C-MRSA may have the advantage over SCCmec I–III carried by H-MRSA because they are smaller and more transferable; both of these putative advantages may contribute to their propagation (10,15).
The close relatedness and high prevalence of this virulent pathogen argue for a clonal expansion advantage of this particular clone. Outbreaks of C-MRSA infections caused by SCCmec IV (IVa) have been reported in several countries (4). Since our genotyping results showed that MRSA isolates possessing SCCmec V and PVL in Taiwan are clonally related, we cannot rule out the possibility of outbreaks due to this particular clone in some areas. However, our isolates came from multiple hospitals throughout the 4 geographic regions of Taiwan. Our data also showed that this particular clone was already present in 2000. In addition, this particular clone was found not only in outpatients but also in ICU and non-ICU inpatients, including in hospital-acquired infections. These findings indicate that this clone has migrated into the hospital environment; moreover, it can cause more severe infections, as shown by its presence in blood, respiratory, ear, and other specimens.
Our analysis of MRSA isolates collected in 2000 and 2002 indicated that a virulent clone of MRSA (pulsotype C:ST59, SCCmec V and PVL-positive), which caused wound infections primarily but also other potentially more serious infections, is highly prevalent in Taiwan inpatient and outpatient settings. Recognition of this clone can be facilitated by its antimicrobial susceptibility profile. Because the resistance pattern of these isolates differs from that of traditional H-MRSA strains, the antimicrobial susceptibility profile has important implications for treatment. Understanding the roles these strains play in MRSA epidemiology helps physicians choose the most appropriate treatment. Prompt and judicious management and infection control measures should help deter further spread of this virulent pathogen.
Dr Feng-Jui Chen is a microbiologist at the National Health Research Institutes, Taiwan. His primary research interest is molecular microbiology, particularly the mechanisms of quorum sensing in S. aureus.
Acknowledgment
We thank Linda McDougal for sharing her expertise in pulsed-field gel electrophoresis typing and the hospitals that participated in TSAR in 2000 and 2002.
References
- Chambers HF. The changing epidemiology of Staphylococcus aureus? Emerg Infect Dis. 2001;7:178–82. DOIPubMedGoogle Scholar
- Vandenesch F, Naimi T, Enright MC, Lina G, Nimmo GR, Heffernan H, Community-acquired methicillin-resistant Staphylococcus aureus carrying Panton-Valentine leukocidin genes: worldwide emergence. Emerg Infect Dis. 2003;9:978–84.PubMedGoogle Scholar
- Gillet Y, Issartel B, Vanhems P, Fournet JC, Lina G, Bes M, Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet. 2002;359:753–9. DOIPubMedGoogle Scholar
- Zetola N, Francis JS, Nuermberger EL, Bishai WR. Community-acquired methicillin-resistant Staphylococcus aureus: an emerging threat. Lancet Infect Dis. 2005;5:275–86. DOIPubMedGoogle Scholar
- Wang CC, Lo WT, Chu ML, Siu LK. Epidemiological typing of community-acquired methicillin-resistant Staphylococcus aureus isolates from children in Taiwan. Clin Infect Dis. 2004;39:481–7. DOIPubMedGoogle Scholar
- Hiramatsu K, Cui L, Kuroda M, Ito T. The emergence and evolution of methicillin-resistant Staphylococcus aureus. Trends Microbiol. 2001;9:486–93. DOIPubMedGoogle Scholar
- Ito T, Katayama Y, Asada K, Mori N, Tsutsumimoto K, Tiensasitorn C, Structural comparison of three types of staphylococcal cassette chromosome mec integrated in the chromosome in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 2001;45:1323–36. DOIPubMedGoogle Scholar
- Ma XX, Ito T, Tiensasitorn C, Jamklang M, Chongtrakool P, Boyle-Vavra S, Novel type of staphylococcal cassette chromosome mec identified in community-acquired methicillin-resistant Staphylococcus aureus strains. Antimicrob Agents Chemother. 2002;46:1147–52. DOIPubMedGoogle Scholar
- McDougal LK, Steward CD, Killgore GE, Chaitram JM, McAllister SK, Tenover FC. Pulsed-field gel electrophoresis typing of oxacillin-resistant Staphylococcus aureus isolates from the United States: establishing a national database. J Clin Microbiol. 2003;41:5113–20. DOIPubMedGoogle Scholar
- Ito T, Ma XX, Takeuchi F, Okuma K, Yuzawa H, Hiramatsu K. Novel type V staphylococcal cassette chromosome mec driven by a novel cassette chromosome recombinase, ccrC. Antimicrob Agents Chemother. 2004;48:2637–51. DOIPubMedGoogle Scholar
- McDonald LC, Lauderdale TL, Shiau YR, Chen PC, Wang HY, Lai JF, The status of antimicrobial resistance in Taiwan among gram-positive pathogens: the Taiwan Surveillance of Antimicrobial Resistance (TSAR) Program, 2000. Int J Antimicrob Agents. 2004;23:362–70. DOIPubMedGoogle Scholar
- National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 6th ed. Approved standard. M7-A6. Wayne (PA): The Committee; 2003.
- Enright MC, Day NP, Davies CE, Peacock SJ, Spratt BG. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol. 2000;38:1008–15.PubMedGoogle Scholar
- Carleton HA, Diep BA, Charlebois ED, Sensabaugh GF, Perdreau-Remington F. Community-adapted methicillin-resistant Staphylococcus aureus (MRSA): population dynamics of an expanding community reservoir of MRSA. J Infect Dis. 2004;190:1730–8. DOIPubMedGoogle Scholar
- Okuma K, Iwakawa K, Turnidge JD, Grubb WB, Bell JM, O'Brien FG, Dissemination of new methicillin-resistant Staphylococcus aureus clones in the community. J Clin Microbiol. 2002;40:4289–94. DOIPubMedGoogle Scholar
Figures
Cite This ArticleTable of Contents – Volume 11, Number 11—November 2005
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Tsai-Ling Lauderdale, Division of Clinical Research, National Health Research Institutes, 35 Keyan Rd, Zhunan Town, Miaoli County 350, Taiwan, Republic of China; fax: 886-37-586-457
Top