Volume 11, Number 12—December 2005
Research
Echinococcosis in Tibetan Populations, Western Sichuan Province, China
Abstract
We screened 3,199 people from Shiqu County, Sichuan Province, China, for abdominal echinococcosis (hydatid disease) by portable ultrasound combined with specific serodiagnostic tests. Both cystic echinococcosis (CE) (Echinococcus granulosus infection) and alveolar echinococcosis (AE) (E. multilocularis) were co-endemic in this area at the highest village prevalence values recorded anywhere in the world: 12.9% were infected with one or the other form (6.8% CE and 6.2% AE). Prevalences of both CE and AE were significantly higher in female than male patients and increased with the age of the person screened. Pastoral herdsmen were at highest risk for infection (prevalence 19.0%). Prevalence of CE varied in 5 townships from 0% to 12.1%, whereas AE prevalence ranged from 0% to 14.3%. Risk factors associated with both infections included the number of owned dogs, frequency of contact with dogs, and sources of drinking water.
Human cystic echinococcosis (CE), caused by infection with the larval stage of Echinococcus granulosus, and alveolar echinococcosis (AE), caused by infection with the larval stage of E. multilocularis, are 2 of the most pathogenic zoonotic parasitic helminthic infections of humans in the Northern Hemisphere (1). Human CE occurs worldwide in association with herding, within which the main dog-sheep cycle for E. granulosus is transmitted (1). Human AE is a much rarer parasitic infection; transmission occurs in several regions of the Northern Hemisphere, including the United States, Europe, Central Asia, Siberia, Japan, and China (2). In China, echinococcosis occurs mainly in western regions and provinces, including Xinjiang Uygur Autonomous Region, Qinghai Province, Gansu Province, Ningxia Hui Autonomous Region, and Sichuan Province (3). A previous pilot survey showed that human echinococcosis was prevalent in western Sichuan Province, situated on the eastern Tibetan Plateau, and that both human CE and AE were present. The average prevalence was 4.0%; CE accounted for 2.1% and AE 1.9% (4).
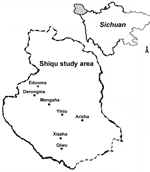
Shiqu County (longitude 97°20´00´´–99°15´28´´E and latitude 32°19´28´´–34°20´40´´N) is located in the northwest corner of Ganzi Prefecture in Sichuan Province (average altitude 4,200 m). The county covers 25,141 km2, located on the eastern part of the Tibetan Plateau. Grassland covers 83.5% of this treeless area, where the weather is cold (annual average temperature –1.6°C). Ethnic Tibetans comprise 98% of the total population; they are primarily involved with livestock production and herding. The total number of livestock is >630,000. In addition, a large number of dogs, including owned dogs and strays, exist in the area (5). We conducted a village-based community epidemiologic study of human echinococcosis from 2000 to 2002 in Shiqu County, Ganzi Tibetan Autonomous Region, Sichuan Province, to further understand the epidemiology of human AE in this region.
The screening program was undertaken from 2000 to 2002; 26 villages in 5 townships in Shiqu County, were included (Figure 1). A total of 3,199 volunteers were self-selected after the purpose of the study was explained to the communities by local village leaders; volunteers were assured free diagnosis and chemotherapeutic treatment for echinococcosis, if indicated. Study participants ranged in age from 1 to 86 years (median 32 years). Fifty-two percent (1,660) were female patients, and 48% (1,539) were male patients. Persons of Tibetan ethnicity comprised 95% of the sampled population. The other participants listed their ethnicity as Han (4.5%), Hui (0.2%), or other (0.3%). Almost half of the participants (52.9%) raised livestock, including yaks, sheep, or goats, as the primary source of their income. Other listed occupations included student (19.1%), public servant (9.8%), preschooler (3.2%), illiterate child (2.0%), semifarmer (2.5%), farmer (1.1%), employee (2.2%), or other (7.3%).
Questionnaire
For each registered participant, a questionnaire designed to obtain information on demographics and animal ownership was completed. Questions mainly concerned occupation, education level, dog ownership and number, frequency of dog contact, fox hunting, drinking water source, and hygienic practices.
Screening and Diagnostic Criteria for Echinococcosis
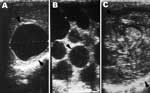
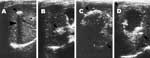
All participants were examined by abdominal ultrasound; those with space-occupying lesions in the liver were asked to give venous blood samples to detect Echinococcus antibody by using enzyme-linked immunosorbent assay (ELISA) and immunoblot with E. granulosus hydatid cyst fluid as antigen (6–8), as well as specific antibodies against E. multilocularis using ELISA and immunoblot with recombinant Em18 antigen (9,10). Diagnosis of human echinococcosis is mainly dependent on pathognomonic ultrasound images complemented by serum antibody confirmation of suspect CE/AE images (6,11). Investigators used the criteria for classification proposed by the World Health Organization Informal Working Group on Echinococcosis for CE (11), and the PNM system for classification of human AE, in which P stands for hepatic location of the parasite, N refers to extrahepatic involvement of neighboring organs, and M stands for absence or presence of distant metastases (12). CE Cases were defined as follows: 1) presence of characteristic cystlike images detected on abdominal ultrasound and a positive ELISA result with hydatid cyst fluid antigen; 2) presence of pathognomonic cyst images detected on abdominal ultrasound, but negative by ELISA (Figure 2). In addition, CE cases, on the basis of the conformational features of cysts, were differentiated into 6 types (CL, CE1, CE2, CE3, CE4, and CE5 ) and subdifferentiated by size into 3 subtypes (small [s], medium [m], and large [l]) within each type. A case of AE was defined as follows: 1) presence of pathognomonic progressive AE type lesion detected on abdominal ultrasound, regardless of serologic results; 2) presence of calcified lesions, 1–3 cm in diameter, or nodular hyperechoic lesions detected on abdominal ultrasound and seropositive against recombinant Em18; and 3) presence of a calcified lesion (1–3 cm in diameter) detected by abdominal ultrasound and negative for antibodies to the recombinant Em18 antigen but positive by ELISA, with hydatid cyst fluid (Figure 3).
Statistical Analysis
All analyses were performed by using EpiInfo version 5.01a (Centers for Disease Control and Prevention, Atlanta, GA, USA). Statistical significance was set at p<0.01.
In this study, 84 (2.6%) of 3,199 participants examined by abdominal ultrasound scanning were determined to have an intrahepatic mass with a nonhomogeneous hyperechoic structure that contained scattered calcifications, and with irregular, poorly defined edges. A central necrotic cavity with a hypoechoic pseudoliquid structure and irregular borders was observed in 79 (2.5%) additional persons. In 53 of these participants the infiltrative lesions measured >10 cm in diameter and invaded or surrounded vascular structures, biliary structures, or both. In the other 26 persons, the lesions were nodular, were 3–5 cm in diameter at the longest dimension, and had calcifications. Calcified lesions, 1–3 cm in diameter, were observed in 20 persons. Thus, 163 persons were confirmed by ultrasound scanning to have AE infection, and 46 were suspected of having AE. Confirmatory serodiagnostic tests were performed in Japan and China, respectively. Serodiagnosis with the EgCF antigen in ELISA was positive in 93 of 94 persons with typical images of AE, 24 of 25 persons with nodular lesions, and 11 of 20 persons with calcified lesions. Additional serologic testing with the rEm18 antigen in ELISA and immunoblot was positive in 101 of 102 persons with typical images of AE, 16 of 25 with nodular lesions, and 8 of 14 with calcified lesions (Table 1). Therefore, positive confirmative serology in 35 study participants with a suspect AE image of a nodular lesion or calcified lesion indicated infection with AE. Another patient with a suspect AE image of a nodular lesion in the liver refused to give venous blood, so confirmative serologic tests could not be performed on him, and this case was not counted in the AE category. Thus, of 46 study participants with a suspect AE image, 35 were finally diagnosed as having AE. A total of 198 (6.2%) of 3,199 persons studied were determined to be infected with AE on the basis of abdominal ultrasound images and confirmatory serologic results; 15 (38.5%) of 39 infected persons had inactive, or abortive AE lesions. Ninety-five single AE lesions were located in the right hepatic lobe, and 31 were in the left hepatic lobe. Involvement of both right and left hepatic lobes by a single lesion was observed in 17 patients. In 55 cases, >2 distinct foci were observed.
In addition, an ultrasound image of CE in the liver was detected in 216 (6.8%) of 3,199 study participants examined. In 10 cases, ultrasound images showed unilocular, cystic lesions with uniform anechoic content, without visible cyst wall, all <5 cm; they were considered to be type CLs. Images characterized by unilocular, simple cyst with uniform anechoic content and visible cyst wall, some exhibiting a snowflake image (7 images <5 cm, 42 images ranging from 5 to 10 cm, and 26 images >10 cm) were observed in 75 patients; they were determined to be Type CE1(7 CE1s, 42 CE1m, 26 CE1l); In 54 patients, images exhibited multivesicular or multiseptate cysts with a wheel-like appearance; others displayed unilocular cysts with daughter cysts with a honeycomb appearance. Eight of these images were <5 cm, 16 images were 5–10 cm, and 30 images were >10 cm; all of these images belonged to type CE2 (8 CE2s, 16 CE2m, 30 CE2l). In 23 cases, images were characterized by anechoic content with detachment of laminated membrane from the cyst wall, visible as a water-lily design; some had a unilocular cyst containing daughter cysts, but the whole cyst form was less rounded. Five of these cysts were <5 cm, 13 cysts were 5–10 cm, and 5 cysts were >10 cm; all were confirmed to be type CE3 (5 CE3s, 13 CE3m, 5 CE3l). In 48 cases, cysts had hyperechoic degenerative contents without daughter cysts. Seventeen of these cysts were <5 cm, 19 were 5–10 cm, and 12 cysts were >10 cm; these images belonged to type CE4 (17 CE4s, 19 CE4m, 12 CE4). Cysts characterized by thick, calcified walls in an arch-shaped form with a cone-shaped shadow, were observed in 6 cases; 3 had images <5 cm, and 3 had cysts 5–10 cm in size; these were determined to be type CE5 (3 CE5s, 3 CE5m). In 18 cases, >1 cystic lesions were identified in the abdominal cavity in addition to the liver cysts. In 5 cases, additional cysts were found in the spleen; in 3 cases, additional cysts were found in the pelvic cavity; and in l case, a cyst was also found in the kidney. Serologic results in these study participants with CE at ultrasound examination are shown in Table 1. Serodiagnosis using the EgCF antigen in ELISA was negative in 16 of 161 persons with CE; 12 of 123 persons with CE were seropositive with rEm18 by ELISA and immunoblot (Table 1). No mixed infections were observed.
Distribution by Sex and Age
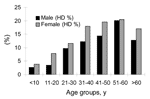
Of 414 persons with evidence of abdominal echinococcosis, 244 (CE = 134, AE = 110) were female patients, and 170 (CE = 82, AE = 88) were male. Thus, the prevalence of echinococcosis in female patients was 14.7% (244/1,660), and 11.0% (170/1,539) in male patients. Thus, prevalence in female patients was significantly higher than in males (χ2 = 9.46, p<0.01). Compared with other older groups, the population <20 years of age had a lower infection prevalence (5.4%). In general, prevalence increased with age and reached a peak in the age group of >50 to <60 (Figure 4). The prevalence in the age group of >10 to <20 years was significantly lower than in the age group of >20 to <30 years (χ2 = 10.20, p<0.01). The youngest person infected with CE was 4 years of age, the oldest one was 79 years, and the average age of persons with CE was 39.0 years (n = 216). The youngest persons with AE was 8 years of age, the oldest 80 years, and the average age of AE patients was 43.1 years (n = 198) (Figure 5).
Village Prevalence
In this study 2,033 persons were screened for echinococcosis from 26 villages in the townships of Yiniu, Mengsha, Arizha, Xiazha, and Qiwu within Shiqu County; 226 infected cases were detected. The overall township prevalence of echinococcosis was 11.1% (range 7.4%–15.1%); 6.2% of patients were diagnosed with AE and 4.9% with CE disease. The highest village prevalences for AE and CE were 14.3% and 12.1%, respectively (Table 2).
Other Risk Factors
Occupation was a major risk factor. Herdsmen had the highest risk for echinococcosis infection, with a total prevalence of 19.0% (322/1,692, p<0.01); the AE prevalence was 9.5% (160/1,692), and the CE prevalence was 9.6% (162/1,692). Part-time herdsmen had a 12.7% prevalence of echinococcosis. Students and preschool children had a lower prevalence (2.8% and 3.0%), while illiterate adolescents were more heavily infected (14.3%) (χ2 = 21.17, p<0.01) (Table 3).
A total of 2,811 of 3,199 persons examined answered the question about dog ownership. Of these, 496 said they did not own dogs; 2,315 (82.4%) persons had various numbers of dogs (range 1–9). Analysis indicated that the population without owned dogs had a total echinococcosis prevalence of 8.3% (41/496) (CE = 4.4%, AE = 3.8%). In contrast, persons who owned dogs had a total echinococcosis prevalence of 15.6% (360/2,315) (CE = 8.0%, AE = 7.5% [Table 4]).
To a certain extent, education can determine occupation choice and lifestyle. Our results implied that prevalence of echinococcosis had some relationship with the level of education. Among herdsmen, 1,469 (86.8%) of 1,692 were illiterate; the prevalence in this subgroup reached 20.0% (293/1,469), the highest rate in the sampled population. The prevalence in self-identified literate herdsmen was 13.0% (29/223). Among illiterate adolescents, 14.3% were infected. Persons with only primary school education had a 6.0% (53/882) combined infection prevalence, and those with middle school education 9.1% (29/318). Persons with university education had an infection rate of 6.3% (17/268), and preschool children had an echinococcosis infection prevalence of 2.9% (3/105).
Fox hunting was also a risk factor. A total of 2,841 of 3,199 persons examined replied to the question about fox hunting. Results showed that the total prevalence of echinococcosis in populations who said that they neither hunted foxes nor kept fox skin products was 7.6% (29/384) (AE = 3.4%, CE = 4.2%), compared to a prevalence of 15.2% (368/2,427) (CE = 7.8% and AE = 7.4%) for persons who said they kept fox skin products that they had purchased, and 10% (3/30) (CE = 3 and AE = 0) in persons who said they kept fox skin products that they obtained by hunting.
In this mass screening study of Tibetan communities, portable ultrasound examination combined with specific serologic tests was used for the diagnosis of both CE and AE. Survey results indicated that human echinococcosis is a serious public health problem for the inhabitants of this area, for whom a 12.9% overall prevalence was recorded. In comparison with reports on human echinococcosis in other areas, including other areas of China, the prevalence in northwest Sichuan Province was much higher for both CE and AE (1,3,12,13). The prevalence of CE was higher than in other recognized echinococcosis-endemic areas of the world, including North Africa, South America, Russia, and the Middle East (1,12,14,15). Previous ultrasound-based surveys for human AE have shown regional prevalences of <0.05% in continental Europe to 4% in Gansu Province in central northwest China (16,17). The most striking observation, however, was that both AE and CE were co-endemic in this area of Sichuan, with a prevalence of 6.8% for CE and 6.2% for AE. Only parts of Turkey, Central Asia, and Siberia have been identified as co-endemic for both human CE and AE (1,14).
In Shiqu County, China, analysis of human CE and AE indicated that prevalence of disease in female patients was significantly higher (14.7%) than in male patients (11.0%). According to traditional Tibetan custom, women are usually responsible for home chores, including feeding dogs, collecting yak dung for fuel, and milking livestock. Thus, women and girls may have more opportunity to be exposed to Echinococcus-infected dogs and the contaminated environment.
The infection prevalence for both CE and AE for persons in the age groups <20 years was markedly lower than those of other age groups. Prevalence reached a peak among the >50- to 60-year age group. The presence of CE or AE in persons as young as 4 and 8 years, respectively, indicates recent active transmission. In general, CE or AE infection increased with age. However, among persons >60 years of age prevalence of both AE and CE declined, a situation consistent with previous reports (4,15,18); this finding may be associated with early death of persons infected with forms of echinococcosis, particularly with AE. A recent analysis of the relative health impact of echinococcosis in these Tibetan communities showed that CE and AE caused an average of 0.8 disability-adjusted life years lost per person (19), which is an exceptional value.
This analysis showed that AE infection varied from 0% to 14.3% by village and that CE village prevalence ranged from 0% to 12.1%. A trend of gradual decrease in AE in villages from north to south (9.4% vs. 0.9% in the 5 townships surveyed) was observed.
Several factors may contribute to the high prevalence of human AE in this Tibetan population. High densities of small mammals are essential to maintaining the transmission cycle of E. multilocularis, and small mammal populations are also subject to ecologic changes, such as deforestation or pasture overgrazing (16,20–22). The involvement of dogs as well as foxes in transmission in eastern Tibet, together with lack of hygiene and probable contamination of the local peridomestic environment, seem to be additional major factors (23,24). For the 5 townships located in the central area of Shiqu County, the geographic conditions, apparent ecologic factors, life style, religion, livestock production, and dog ownership practices appear to be similar; however, human AE village prevalence was markedly variable. We had previously observed that local differences in small mammal abundance over time, possibly associated with overgrazing practices may contribute to variable township AE disease rates (22).
This survey disclosed that 86.8% of herdsmen were illiterate; 20% of them had either CE or AE disease. Consequently, improving the knowledge and awareness of the disease among the traditional nomadic population is imperative in any future control or prevention studies. Analysis indicated that both CE and AE risk was related to dog ownership (p<0.01), contact with dogs (p<0.01), source of drinking water, and general hygiene (p<0.01). While the role of domestic and working dogs as the major definitive host for E. granulosus is clear, such is not the case for E. multilocularis. Of particular interest therefore was the strong association between human AE risk and dog ownership or contact. Evidence from community studies in other parts of China (16), the United States (25), and Germany (26) increasingly show that the domestic dog plays a key role in the zoonotic risk for human AE.
Dogs are kept in large numbers by Tibetans and are used primarily to guard property and livestock. In this survey, 82.4% of the population owned dogs, and 21% owned >3 dogs. Buddhist practice forbids killing any animal, including dogs, and this practice leads to large numbers of stray dogs, which mainly gather around temples or townships, where they are fed by monks and herdsmen. Dogs also are predators of small mammals on adjacent pastures; these dogs are usually fed by herdsman with offal (including liver and lungs) of sheep and yaks during slaughtering season. Necropsy of intestines of stray dogs in 1995 in this region showed a 29.5% prevalence for E. granulosus and 11.5% for E. multilocularis (27,28). A recent diagnostic purgation study of dogs in this area demonstrated E. multilocularis prevalence of 12% and an E. granulosus prevalence of 8% (29). Foxes are the main sylvatic hosts of E. multilocularis, and both the Tibetan fox (Vulpes ferrilata) and the red fox (V. vulpes) are common on the Qinghai-Tibet plateau. A previous report showed a high prevalence of E. multilocularis in the Tibetan fox (59.1%) and red fox (57.1%) (28) in this area. Furthermore, Qiu et al. observed in 1995 the existence of E. strobilae in Tibetan foxes with morphologic characteristics distinct from E. multilocularis adults but considered it to be a variant of E. multilocularis. These specimens and new samples have been shown to be a new species of taeniid cestode belonging to the genus E. Rudolphi (30). However, whether the new species is involved in the transmission of a third form of human echinococcosis in this region has yet to be determined.
Dr Tiaoying is an assistant at the Institute of Parasitic Diseases, Sichuan Centers for Disease Control and Prevention. Her research interests include epidemiologic study, chemotherapy, and serodiagnosis of echinococcosis, particularly ultrasound diagnosis.
Acknowledgment
This study was supported by the Ecology of Infectious Disease Program of the National Institutes of Health and National Science Foundation, USA (1R01 Two1565-01) and by the Sichuan Provincial Department of Health, China.
References
- Schantz PM, Chai JJ, Craig PS, Echert J, Jenkins DJ, Macpherson CNL, Epidemiology and control of hydatid disease. In: Thompson RCA, Lymbery AJ, editors. Echinococcosis and hydatid disease. Wallingford, UK: CAB International; 1995. p. 233–331.
- Kern P, Bardonnet K, Renner E, Auer H, Pawlowski Z, Ammann RW, European echinococcosis registry: human alveolar echinococcosis, Europe, 1982–2000. Emerg Infect Dis. 2003;9:343–9.PubMedGoogle Scholar
- Qiu JM, Liu FJ, Schantz PM, Ito A, Delker C, He JG, Epidemiological study on human hydatidosis in Tibetan region of Western Sichuan. Chin J Zoonoses. 2000;10:77–80.
- The committee of compiling Shiqu County annals. The Shiqu annals. Chengdu, Sichuan Province, China: Sichuan People's Press; 2000. p. 1–3.
- Bartholomot G, Vuitton DA, Harraga S, Snida Z, Giraudoux P, Barnish G, Combined ultrasound and serologic screening for hepatic alveolar echinococcosis in central China. Am J Trop Med Hyg. 2002;66:23–9.PubMedGoogle Scholar
- Ito A, Ma L, Schantz PM, Gottstei B, Liu YH, Chai JJ, Differential serodiagnosis for cystic and alveolar echinococcosis using fractions of Echinococcus granulosus cyst fluid (antigen B) and E. multilocularis protoscolex (Em18). Am J Trop Med Hyg. 1999;60:188–92.PubMedGoogle Scholar
- Mamuti W, Yamasaki H, Sako Y, Nakaya K, Nakao M, Lightowlers MW, Usefulness of hydatid cyst fluid of Echinococcus granulosus developed in mice with secondary infection for serodiagnosis of cystic echinococcosis in humans. Clin Diagn Lab Immunol. 2002;9:573–6.PubMedGoogle Scholar
- Xiao N, Mamuti W, Yamasaki H, Sako Y, Nakao M, Nakaya K, Evaluation of use of recombinant Em18 and affinity-purified Em18 for serological differentiation of alveolar echinococcosis from cystic echinococcosis and other parasitic infections. J Clin Microbiol. 2003;41:3351–3. DOIPubMedGoogle Scholar
- Ito A, Sako Y, Yamasaki H, Mamuti W, Nakaya K, Nakao M, Development of Em18-immunoblot and Em18-ELISA for specific diagnosis of alveolar echinococcosis. Acta Trop. 2003;85:173–82. DOIPubMedGoogle Scholar
- World Health Organization. International classification of ultrasound images in cystic echinococcosis for application in clinical and field epidemiological settings. In PAIR, an option for the treatment of cystic echinococcosis. Geneva: The Organization; 2001. p. 23–9.
- Eckert J, Gemmell MA, Meslin F-X, Pawlowski ZS. WHO/OIE manual on echinococcosis in humans and animals: a public health problem of global concern. Paris: World Health Organization/World Organization for Animal Health; 2001. p. 48–50.
- Wang HL, Yin YC, Ma Z, Zhang CY, Zhang XP, Cheng RP, A preliminary report on investigations of both cystic and alveolar forms of human hydatidosis of the liver in Xihou County, Ningxia Province. Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:143–5.PubMedGoogle Scholar
- Kovalenko F, Darchenkova N, Legonkov Y, Musaev G, Gudovsky L, Parshin V, Hydatid diseases (cystic and alveolar) in Russia (1983–1997). Acta Parasitol. 2000;45:241–2.
- Shambesh MK, Macpherson CN, Beesley WN, Gusbi A, Elsonosi T. Prevalence of human hydatid disease in north-west Libya: a cross-sectional ultrasound study. Ann Trop Med Parasitol. 1992;86:381–6.PubMedGoogle Scholar
- Craig PS, Giraudoux P, Shi D, Bartholomot B, Garnish G, Delattre P, An epidemiological and ecological study of human alveolar echinococcosis transmission in south Gansu, China. Acta Trop. 2000;77:167–77. DOIPubMedGoogle Scholar
- Romig T, Kratzer W, Kimmig P, Frosch M, Gaus W, Flegel WA, An epidemiologic survey of human alveolar echinococcosis in southwestern Germany. Am J Trop Med Hyg. 1999;61:566–73.PubMedGoogle Scholar
- Bchir A, Larouze B, Soltani M, Hamdi A, Bouhaouala H, Ducic S, Echotomographic and serological population-based study of hydatidosis in central Tunisia. Acta Trop. 1991;49:149–53. DOIPubMedGoogle Scholar
- Budke CM, Jiamin Q, Zinsstag J, Qian W, Torgerson PR. Use of disability adjusted life years in the estimation of the disease burden of echinococcosis for a high endemic region of the Tibetan Plateau. Am J Trop Med Hyg. 2004;71:56–64.PubMedGoogle Scholar
- Giraudoux P, Quere JP, Delattre P, Bao G, Wang X, Shi D, Distribution of small mammals along a deforestation gradient in southern Gansu, Central China. Acta Ther. 1998;43:349–62.
- Zhou HX, Chai SX, Craig PS, Delattre P, Quere JP, Raoul F, Epidemiology of alveolar echinococcosis in Xinjiang Uygur autonomous region, China: a preliminary analysis. Ann Trop Med Parasitol. 2000;94:715–29.PubMedGoogle Scholar
- Wang Q, Vuitton DA, Qiu J, Giraudoux P, Xiao Y, Schantz PM, Fenced pasture: a possible risk factor for human alveolar echinococcosis in Tibetan pastoralist communities of Sichuan, China. Acta Trop. 2004;90:285–93. DOIPubMedGoogle Scholar
- Qiu JM, Liu FJ, Wang H, Ito A, Schantz PM. A survey of hydatid disease in Tibetan population in China: correlation of ultrasound and radiologic imaging and serologic result. Archivos Internacionales de la Hidatidosis. 1999;33:211–3.
- Wang Q, Qiu JM, Schantz PM, He JG, Ito A, Liu FJ. Investigation of risk factors for development of human hydatidosis among households raising livestock in Tibetan areas of western Sichuan Province. Chinese Journal of Parasitology and Parasitic Diseases. 2001;19:93–6.PubMedGoogle Scholar
- Rausch RL, Wilson JF, Schantz PM. A programme to reduce the risk of infection by Echinococcus multilocularis: the use of praziquantel to control the cestode in a village in the hyperendemic region of Alaska. Ann Trop Med Parasitol. 1990;84:239–50.PubMedGoogle Scholar
- Kern P, Ammon A, Kron M, Sinn G, Sander S, Petersen LR, Risk factors for alveolar echinococcosis in humans. Emerg Infect Dis. 2004;10:2088–93.PubMedGoogle Scholar
- Qiu JM, Qiu DC, Luo CX, Zhu YB, Chen XW. Survey on infective agent of alveolar echinococcosis in Ganzi Prefecture and experimental research in animal. Chin J Zoonoses. 1989;5:38–40.
- Qiu JM, Chen XW, Ren M, Lu CX. Epidemiological study on alveolar hydatid disease in Qinghai-Xizang Plateau. Journal of Practical Parasitic Diseases. 1995;3:106–8.
- Budke CM, Campos-Ponce M, Qian W, Torgerson PR. A canine purgation study and risk factor analysis for echinococcosis in a high endemic region of the Tibetan Plateau. Vet Parasitol. 2005;127:43–9. DOIPubMedGoogle Scholar
- Xiao N, Qiu J, Nakao M, Li T, Yang W, Chen X, Echinococcus shiquicus n. sp., a taeniid cestode from Tibetan fox and plateau pika in China. Int J Parasitol. 2005;35:693–701. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 11, Number 12—December 2005
| EID Search Options |
|---|
|
|
|
|
|
|
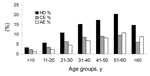
Please use the form below to submit correspondence to the authors or contact them at the following address:
Qiu Jiamin, Department of Echinococcosis and Cysticercosis Control, Institute of Parasitic Diseases, Sichuan Centers for Disease Control and Prevention, Chengdu 610041, Sichuan Province, People’s Republic of China; fax: 86-28-5438409
Top