Volume 12, Number 6—June 2006
Research
Genetic Divergence of Toxoplasma gondii Strains Associated with Ocular Toxoplasmosis, Brazil
Abstract
Previous studies have shown a high prevalence of toxoplasmosis and the frequent occurrence of ocular disease in Brazil. To identify the genotypes of parasite strains associated with ocular disease, we compared 25 clinical and animal isolates of Toxoplasma gondii from Brazil to previously characterized clonal lineages from North America and Europe. Multilocus nested polymerase chain reaction analysis was combined with direct sequencing of a polymorphic intron to classify strains by phylogenetic methods. The genotypes of T. gondii strains isolated from Brazil were highly divergent when compared to the previously described clonal lineages. Several new predominant genotypes were identified from different regions of Brazil, including 2 small outbreaks attributable to foodborne or waterborne infection. These findings show that the genetic makeup of T. gondii is more complex than previously recognized and suggest that unique or divergent genotypes may contribute to different clinical outcomes of toxoplasmosis in different localities.
Toxoplasma gondii is an obligate intracellular parasite that infects a wide range of warm-blooded vertebrates and causes disease in agricultural animals and humans (1). T. gondii has a complex life cycle that includes an asexual cycle and sexual cycle; the asexual cycle occurs in a wide range of intermediate hosts, and the sexual cycle occurs exclusively in feline hosts, which shed infectious oocysts in their feces (1). T. gondii is mainly transmitted by ingesting cysts contained within tissues of a chronically infected host or by ingesting sporulated oocysts from fecally contaminated food or water (2). T. gondii is an influential foodborne pathogen in the United States (3) and a frequent cause of waterborne infection in parts of Brazil (4,5).
Despite having a sexual phase in its life cycle, the population structure of T. gondii is markedly clonal (6). Most strains analyzed from North America and Europe belong to 1 of 3 clonal lineages known as types I, II, and III (7–9). A small number (<5%) of isolates contain different combinations of the same alleles seen in the clonal types, which indicates that recombination occurs infrequently in the wild (7). Additionally, strains with more divergent genotypes have been isolated from locations such as French Guiana (10).
The serologic prevalence of T. gondii infection in Brazil includes 50%–80% of the adult population, with the highest values found in northern and southern states (5). Waterborne transmission has been implicated in high rates of T. gondii seropositivity in northern Rio de Janeiro State (5) and in a toxoplasmosis outbreak in Santa Isabel do Ivai in southern Paraná (4). High levels of ocular disease are associated with toxoplasmosis in Brazil (11). In the southern city of Erechim, Brazil, 184 (17.7%) of 1,042 adults were found to have retinal scars, thought to be caused by toxoplasmosis (12). Epidemiologic data indicate that many cases of ocular disease are acquired after birth rather than congenitally (13–15). Whether the increased prevalence and severity of ocular toxoplasmosis in Brazil are attributable to host or parasite genetic factors or differences in exposure rate is uncertain.
Polymerase chain reaction (PCR)–based typing at the SAG2 locus has been used previously to suggest that type I strains predominate in Brazilian patients with ocular toxoplasmosis (16). While the SAG2 marker provides accurate genotyping for most strains within the clonal lineages, it cannot detect recombinant strains or those with unusual genotypes (17). In fact, the exclusive use of any single locus may misrepresent the genotype of recombinant or unusual genotypes as having a simple genotype. This problem is partially alleviated by multilocus analysis, and random amplified polymorphic DNA (RAPD)–PCR analysis of Brazilian T. gondii strains with multiple markers showed that most strains contain both type I and III alleles at different loci (18). However, PCR-based markers, such as restriction fragment length polymorphism (RFLP) or RAPD, underestimate the true rate of nucleotide divergence and thus may not accurately classify T. gondii strains from new regions. For example, a high degree of polymorphism is detected at the GRA6 locus by sequence analysis (9 allelic sequences among 30 strains), whereas PCR-RFLP analysis differentiates only 3 groups among these same strains (19).
We have recently described a sensitive method for multilocus genotyping consisting of nested PCR (nPCR) amplification of 4 different RFLP markers (SAG2, GRA6, SAG3, and BTUB). When combined with sequencing of the UPRT-1 intron, multilocus nPCR typing provides a robust means to classify strains as having clonal, recombinant, or novel genotypes (17). Multilocus nPCR analysis also can detect as few as 5 parasite genomes and thus is applicable to low-volume samples containing few parasites, as is typical of clinical specimens (17). In this study, we examined a group of Brazilian T. gondii strains from animal and human sources, including several outbreaks, to examine the population structure of T. gondii in Brazil.
Clinical Isolates
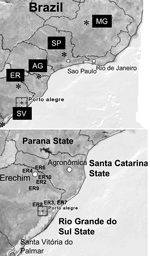
Patients were examined at Clinica Silveira, Erechim, Rio Grande do Sul State, by indirect ophthalmoscopy or biomicroscopy by using a slit-lamp microscope. Ocular disease was evaluated on the basis of parameters described previously (11). Patient consent was obtained at the time of sample acquisition. Recently acquired toxoplasmosis was confirmed by serologic tests that monitor immunoglobulin G (IgG) and IgM by enzyme-linked immunosorbent assay (Abbott Laboratories, Abbott Park, IL, USA) (11). Ocular toxoplasmosis was diagnosed on the basis of recurrent episodes of necrotizing retinochoroiditis. Venous blood was collected before treatment in Vacutainer tubes containing heparin and cells, and serum was separated by centrifugation. The buffy-coat layer was removed, frozen at –20°C, and shipped to Washington University, Saint Louis, for analysis. Blood was also obtained from patients infected during 2 small outbreaks of toxoplasmosis in Santa Vitoria do Palmar, Rio Grande do Sul State, and Agronomica, Santa Catarina State, and processed in a similar manner. Buffy-coat samples were processed by using the DNAeasy tissue extraction kit (Qiagen Inc., Valencia, CA, USA) before PCR analysis.
Parasite Strains and Tissue Samples
Reference strains consisted of representative members of the 3 clonal lineages originally isolated from human or animal infections in North America and Europe. Reference strains for the clonal types included: 4 type I strains: ENT (ATCC 50850), RH (ATCC 50838), GT1, and VEL (ATCC 50852); 3 type II strains: Me49 (ATCC 50840), DEG (ATCC 50855), and PIH (ATCC 50857); and 3 type III strains: CTG (ATCC 50842), STRL, and VEG (ATCC 50861). In addition, 3 previously reported strains with more divergent genotypes were included: CAST (ATCC 50868), COUG, and MAS (ATCC 50870) (20). Parasite strains were grown in human fibroblast cells, harvested after natural egress from host cells and purified as above; cell lysates were prepared for PCR as described previously (17). Five Brazilian T. gondii strains isolated from Belo Horizonte, Minas Gerais, and one from São Paulo, as described previously (18), were also included.
Additional samples of porcine tissue collected from abattoirs in the Erechim region were included (R.N. Belfort, unpub. data). Six samples were analyzed, 2 negative controls and 4 samples that were positive by PCR (data not shown). Tissue samples were extracted with DNAzol, followed by an equal volume of chloroform. Polyacryl carrier (Molecular Research Center, Inc., Cincinnati, OH, USA) was added (5 μL) to the aqueous phase, and DNA was precipitated by adding an equal volume of ethanol and centrifugation at 5,000 × g for 10 min.
Genotyping Isolates by PCR-RFLP
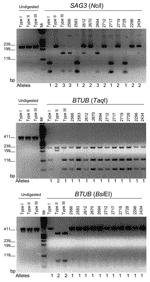
Multilocus nPCR analysis of 4 different loci was based on the markers 5´-SAG2, 3´-SAG2, BTUB, GRA6, and SAG3 (17). Amplification was performed as described previously (20), and negative controls consisted of sterile, distilled water or proteinase K–treated cell lysate of noninfected host cells. The amplified products were digested with appropriate restriction enzymes for different loci, and the resulting fragments were analyzed by 3% agarose gel electrophoresis, stained with ethidium bromide, and imaged by an Alpha Imager version 5.5 camera (Alpha Innotech Corp., San Leandro, CA, USA).
Restriction fragments were scored visually as present or absent, and a genetic distance matrix was calculated from the proportion of shared restriction sites by using the equation of Nei and Li (21). The neighbor-joining method was used to analyze the distance matrix, and dendrograms were generated by using the phylogenetic analysis program PAUP* version 4.0b (22). Bootstrap analysis was conducted for 1,000 replicates to obtain confidence estimates for the taxonomic groupings. The conditions were set to distance, neighbor-joining, with mean character differences, and dendrograms were constructed by using the 50% majority rule.
UPRT-1 Intron Sequences Analysis
Sequence divergence among strains of T. gondii was determined at the uracil phosphoribosyl transferase (UPRT) intron 1 sequence (GenBank accession no. AY143141), as described previously (17). Following the previously described nPCR amplification of the UPRT-1 intron, a third set of internal primers was used for sequencing: UPRT-1seqF 5´-CTCGTCCTCGTTTTCCTT-3´ and UPRT-1seqR 5´-TGAAAGGAAGCACGTAAAGT-3´. Sequencing was conducted on 3 independent PCR-amplified templates by using BigDye cycle sequencing (Applied Biosystems, Foster City, CA, USA) (conducted by SeqWright DNA Technology Services, Houston, TX, USA). ClustalX/W (23) was used to align the sequences for comparison with default settings. After removal of primer sequences, the UPRT-1 intron sequence used for comparison was 467 bp in length. Unrooted phylogenetic comparisons were conducted with distance and parsimony methods by using PAUP*4.0b (22). The conditions were set to distance (mean character difference, minimal evolution, negative branches = 0), and 1,000 bootstrap replicates were performed by using the BioNeighbor-Joining algorithm. Alternatively, parsimony analysis was conducted by heuristic stepwise searching, with bootstrapping for >1,000 replicates. Consensus trees were drawn with an arbitrary root according to the bootstrap 50% majority rule.
Brazil has a high prevalence of ocular toxoplasmosis, and many of these cases are recurrent and serious in nature (11,12). This situation prompted us to consider whether sampling patient blood might allow diagnosis of recent (acute) or recurrent infection by direct PCR amplification. Blood was collected from 77 patients seen at the Clinica Silveira, Erechim, Brazil, from 2003 to 2005, and the buffy-coat that contained leukocytes was separated by centrifugation and used for analysis. Nested PCR analysis of these samples by using the SAG3 gene showed that 11 of 77 were positive, including 6 patients with acute disease and 5 patients with recurrent disease (Table) (locations of the patients with positive samples are shown in Figure 1). We also analyzed several sets of samples from 2 small outbreaks of acute toxoplasmosis. The first in Santa Vitoria do Palmar consisted of 10 persons from a single family that shared a meal of home-cured sausage that contained pork. Symptoms in infected persons included lymphadenitis, myalgia, fever, headache, and sweating. One of these patients, a 53-year-old woman, had severe retinochoroiditis. Only a single sample from these 10 persons was positive by SAG3 nPCR analysis of buffy-coat cells. A second outbreak consisted of 8 infected persons from Agronomica, a town of ≈4,000 residents located 200 km from Florianopolis. These 8 persons shared the same source of nontreated water in a common neighborhood, and their illness was likely caused by waterborne infection. Three of these persons had positive results by SAG3 nPCR.
To determine the genotype of T. gondii strains present in clinical samples from Brazil, multilocus nPCR was conducted by using 4 independent markers, SAG2, BTUB, GRA6, and SAG3, as described previously (17). The 15 clinical samples found to be positive for SAG3 were genotyped for most of these markers, although in some cases, insufficient material was available to type all markers (Table). We compared these isolates to strains previously characterized from Brazil and to the clonal lineages common in North America and Europe. In total, 38 strains were subjected to multilocus nPCR analysis, and after restriction digestion and gel electrophoresis of the products, the strains were classified on the basis of the alleles present relative to the reference strains (Table, Figure 2) (17). Three of the ocular toxoplasmosis samples carried alleles characteristic of type I strains at 3 or more independent markers, and 2 Brazilian chicken strains possessed alleles typical of type III strains at all loci. All of the remaining Brazilian samples had genotypes consisting of different combinations of alleles seen in the clonal types. Nine clinical samples, including samples from Agronomica from 2 outbreaks, possessed the same profile that consisted of alleles typical of type I and type II lineages. The nPCR assay used here can detect both alleles equally well for all the makers studied, yet in no case were 2 alleles detected at a single locus within a single strain (data not shown). Consequently, the genotypes observed in Brazilian isolates cannot be explained by "mixtures" of >1 strain in a given patient or sample.
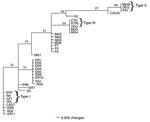
The percentage of nucleotide divergence between strains was estimated from the proportion of shared restriction sites at each locus, and a distance matrix was used to construct a dendrogram by using neighbor-joining analysis (Figure 3). All the strains belonging to type II were clustered together with a high degree of confidence. All type III strains and 2 chicken strains from Brazil were grouped together with a similarly high confidence. The 4 type I reference strains and 3 human Brazilian clinical isolates (ER8, ER11, and AG2) were clustered together. However, most Brazilian T. gondii strains were clustered into 2 new groups that were intermediate between types I and III. These results suggest the presence of at least 2 additional haplotypes that are prevalent in Brazil and which differ from North America and European lineages. MAS, which was isolated from a patient with a congenital case of toxoplasmosis in France, clustered with 1 of these Brazilian haplotypes.
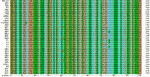
To more accurately assess genetic divergence, we characterized the strains by UPRT-1 intron sequencing, a method that is highly sensitive for detecting divergent strains (17). UPRT-1 intron sequences from 35 strains (1 clinical sample was not available in sufficient quantity for analysis, and 2 samples gave unsatisfactory sequence quality) were aligned by using Clustal X (Figure A1), and the relative divergence of different Brazilian strains was determined by phylogenetic comparison. The results of parsimony and distance analysis were similar and the neighbor-joining distance analysis is shown in Figure 4. Because of the strongly biallelic pattern of T. gondii, types II and III are identical at the UPRT locus, while type I possess a unique haplotype distinguished by 6 single nucleotide polymorphisms (17,20). Most Brazilian T. gondii strains (13 of 22) shared a new allele that was distinguished by 6 additional polymorphisms not seen in the clonal lineages (Figure A1). This new Brazilian allele was also shared by the previously characterized divergent strain MAS (Figure 4). Additionally, 3 outbreak strains (AG1, AG2, AG3) from Agronomica and 1 strain each from chickens (MG4) and pigs (P3) were found in this group that otherwise contained a majority of ocular toxoplasmosis isolates from the Erechim region. Other strains from Brazil contained equally divergent but unique alleles that in some cases formed smaller groups (i.e., P1, P2, P4 and MG1, ER4) (Figure 4). Only a single Brazilian strain (ER8) contained a haplotype characteristic of 1 of the clonal lineages, and this strain was identical to the type I lineage in both the PCR-RFLP and UPRT-1 intron trees.
T. gondii is highly prevalent in Brazil, where human infection is associated with an unusually high occurrence of ocular disease in some locations. We examined the genotype of T. gondii strains collected from a variety of sources in southern Brazil. Included in this study were a group of patients seen at an eye clinic in Erechim, a region known for high levels of ocular toxoplasmosis (11). We also examined strains from several small outbreaks from nearby regions and compared these strains to animal isolates from Erechim and the more central region of Minas Gerais. Multilocus PCR-RFLP and sequenced-based analysis showed that they differ substantially from the previously described clonal lineages and instead define several new haplotypes that appear to be predominant in Brazil. The abundance of genotypes that do not fit the conventional classification shows the global pattern of T. gondii population structure to be more complex than previously thought. These findings have implications for the transmission of T. gondii as a waterborne and foodborne human pathogen and for studies on the role of genetic composition in virulence, pathogenesis, and life cycle dynamics.
Most human infections of T. gondii are not clinically severe and progress rapidly to a chronic state that is characterized by semidormant tissue cysts (2,24). During the chronic infection, parasites are generally not found in circulation, and obtaining parasites without performing invasive procedures such as tissue biopsy is relatively difficult. Previous reports have suggested that parasites may be found circulating in blood during reactivation of toxoplasmosis in AIDS patients (25,26). Our studies show that by using highly sensitive and specific nPCR, small numbers of parasites may be detected in circulating blood from some patients with either acute-onset or recurrent ocular toxoplasmosis. False-positive PCR amplification did not appear to be a substantial problem, as shown by consistently negative results for water and host-cell-only samples and the fact that the genotypes of clinical strains did not resemble common laboratory strains that would be the likely source of any contamination.
Genotyping T. gondii strains found in clinical and animal samples from Brazil showed that all strains except 1 (ER8) had different genotypes from clonal lineages that predominate in North America and Europe. When analyzed by multilocus PCR-RFLP, these new South American genotypes initially appeared to be composed of different combinations of alleles seen in the clonal types, similar to findings of a previous report from Brazil (18). This pattern could indicate that Brazilian strains of T. gondii undergo more frequent sexual recombination, resulting in mixed genotypes. However, the true extent of sequence divergence is not captured by multilocus RFLP analysis. We have previously shown that direct sequencing of introns from housekeeping genes provides a more accurate picture of sequence divergence (17,20). Introns are also likely to be selectively neutral and therefore well suited for phylogenetic comparisons (27). In the present study, when the UPRT-1 intron sequence was compared, all strains from Brazil except 1 (ER8) had multiple additional polymorphisms not seen in the clonal lineages. This locus indicates a low genetic diversity in T. gondii strains in Brazil, although they include genotype(s) uncommon in North America and Europe. Both the RFLP and intron analysis indicate several predominant haplotypes in Brazil, along with less common unique genotypes. Further studies will be necessary to define the population structure of T. gondii in Brazil and other South American locations.
The high seropositivity to T. gondii (11,28,29), combined with unusually high levels of ocular disease in some regions, shows that toxoplasmosis is a notable health problem in Brazil. Previous studies have shown a high prevalence of T. gondii in food animals such as pigs (30) and chickens (31) and in companion animals such as dogs (32) and cats (33). Although companion animals are not typically a source of human infection, the high prevalence in these species indicates a high level of transmission in Brazil. A recent survey of pig samples obtained from abattoirs in the Erechim region indicated a high prevalence of T. gondii (35%–66% positive by PCR) (R.N. Belfort, unpub. data). Previous studies have shown a high level of recurrent ocular disease from this region, where 17.7% adults were found to have retinal scars, likely due to toxoplasmosis (11,12). In addition, drinking unfiltered water has been associated with an increased risk of T. gondii seropositivity in north Rio de Janeiro State, Brazil (5).
Collectively, these epidemiologic features suggest that infection with T. gondii in Brazil is more likely to lead to serious ocular disease, even in otherwise healthy persons. The extent to which host genetics, immune status, and exposure rate contribute to this pattern is unknown. However, an obvious difference is the markedly different genetic makeup of Brazilian strains of T. gondii. Previous studies of recurrent ocular toxoplasmosis in patients in the United States have also shown an elevated frequency of unusual genotypes (34). Although small animal models have been used for evaluating virulence traits of T. gondii strains (6), comparisons have not yet been made between North American and South American strains in terms of their potential to cause ocular disease.
We have previously advocated using SAG2 for genotyping T. gondii strains, since it is capable of distinguishing all 3 clonal genotypes at a single locus (35). This approach works well in North America and Europe, where the 3 major lineages predominate because of extreme linkage disequilibrium (7). Our current findings indicate that most strains from Brazil do not fit the clonal pattern seen in North America. Additionally, T. gondii strains isolated from French Guiana are also genetically distinct from the clonal lineages seen in North America (10). Consequently, studies that rely solely on SAG2 typing will necessarily underrepresent the true genetic divergence in many regions. SAG2 typing has been used for genotyping T. gondii isolates from various animals in Brazil (30–33,36), other parts of South America (37,38), and Africa (39,40). Researchers also recently suggested that strains associated with an outbreak of waterborne toxoplasmosis in Paraná, Brazil, were type I strains, based solely on genotyping with the SAG2 marker (4). However, analyses based solely on SAG2 almost certainly underestimate the genetic diversity of T. gondii in these regions. Further strain comparisons based on a wider set of sequence-based markers will be necessary to define the global population structure of T. gondii and to resolve the relationships between major strain types seen in different regions. Establishing the population structure of T. gondii is highly relevant to transmission dynamics because the suggestion has been made that recently derived clonal lineages arose through a process of recombination that led to enhanced asexual oral transmission (20). Whether other, more divergent strains also express this trait and to what extent their genetic makeup contributes to transmission are highly relevant to understanding the pathogenesis of toxoplasmosis.
Dr Khan is a postdoctoral research associate at Washington University. His research interests include molecular epidemiology and microbial pathogenesis.
Acknowledgments
We thank J.P. Dubey, Tovi Lehmann, Chunlei Su, Ben Rosenthal, Michael Grigg, Jim Ajioka, and Blima Fux for helpful comments and discussions and Julie Suetterlin for expert technical assistance.
This study was supported by a grant from the National Institutes of Health (AI059176).
References
- Dubey JP, Beattie CP. Toxoplasmosis of animals and man. Boca Raton (FL): CRC Press; 1988. p. 220.
- Mead PS, Slutsker L, Dietz V, McCaig LF, Bresee JS, Shapiro C, Food-related illness and death in the United States. Emerg Infect Dis. 1999;5:607–25. DOIPubMedGoogle Scholar
- Moura L, Bahia-Oliveira LMG, Wada MA, Jones JL, Tuboi SH, Carmo EH, Waterborne toxoplasmosis, Brazil, from field to gene. Emerg Infect Dis. 2006;12:326–9.PubMedGoogle Scholar
- Bahia-Oliveira LM, Jones JL, Azevedo-Silva J, Alves CC, Orefice F, Addiss DG. Highly endemic, waterborne toxoplasmosis in north Rio de Janeiro state, Brazil. Emerg Infect Dis. 2003;9:55–62.PubMedGoogle Scholar
- Sibley LD, Boothroyd JC. Virulent strains of Toxoplasma gondii comprise a single clonal lineage. Nature. 1992;359:82–5. DOIPubMedGoogle Scholar
- Howe DK, Sibley LD. Toxoplasma gondii comprises three clonal lineages: correlation of parasite genotype with human disease. J Infect Dis. 1995;172:1561–6. DOIPubMedGoogle Scholar
- Dardé ML, Bouteille B, Pestre-Alexandre M. Isoenzyme analysis of 35 Toxoplasma gondii isolates and the biological and epidemiological implications. J Parasitol. 1992;78:786–94. DOIPubMedGoogle Scholar
- Ajzenberg D, Cogne N, Paris L, Bessieres MH, Thulliez P, Fillisetti D, Genotype of 86 Toxoplasma gondii isolates associated with human congenital toxoplasmosis and correlation with clinical findings. J Infect Dis. 2002;186:684–9. DOIPubMedGoogle Scholar
- Ajzenberg D, Bañuls AL, Su C, Dumètre A, Demar M, Carme B, Genetic diversity, clonality and sexuality in Toxoplasma gondii. Int J Parasitol. 2004;34:1185–96. DOIPubMedGoogle Scholar
- Silveira C, Belfort R Jr, Muccioli C, Abreu MT, Martins MC, Victora C, A follow-up study of Toxoplasma gondii infection in southern Brazil. Am J Ophthalmol. 2001;131:351–4. DOIPubMedGoogle Scholar
- Glasner PD, Silveira C, Kruszon-Moran D, Martins MC, Burnier M, Silveira S, An unusually high prevalence of ocular toxoplasmosis in southern Brazil. Am J Ophthalmol. 1992;114:136–44.PubMedGoogle Scholar
- Gilbert RE, Stanford MR. Is ocular toxoplasmosis caused by prenatal or postnatal infection? Br J Ophthalmol. 2000;84:224–6. DOIPubMedGoogle Scholar
- Holland GN. Reconsidering the pathogenesis of ocular toxoplasmosis. Am J Ophthalmol. 1999;128:502–5. DOIPubMedGoogle Scholar
- Holland GN. Ocular toxoplasmosis: a global reassessment. Part I: epidemiology and course of disease. Am J Ophthalmol. 2003;136:973–88. DOIPubMedGoogle Scholar
- Vallochi AL, Muccioli C, Martins MC, Silveira C, Belfort R Jr, Rizzo LV. The genotype of Toxoplasma gondii strains causing ocular toxoplasmosis in humans in Brazil. Am J Ophthalmol. 2005;139:350–1. DOIPubMedGoogle Scholar
- Khan A, Su C, German M, Storch GA, Clifford D, Sibley LD. Genotyping of Toxoplasma gondii strains from immunocompromised patients reveals high prevalence of type I strains. J Clin Microbiol. 2005;43:5881–7. DOIPubMedGoogle Scholar
- Ferreira AM, Vitor RW, Carneiro ACAV, Brandão GP, Melo MN. Genetic variability of Brazilian Toxoplasma gondii strains detected by random amplified polymorphic DNA-polymerase chain reaction (RAPD-PCR) and simple sequence repeat anchored-PCR (SSR-PCR). Infect Genet Evol. 2004;4:131–42. DOIPubMedGoogle Scholar
- Fazaeli A, Carter PE, Dardé ML, Pennington TH. Molecular typing of Toxoplasma gondii strains by GRA6 gene sequence analysis. Int J Parasitol. 2000;30:637–42. DOIPubMedGoogle Scholar
- Su C, Evans D, Cole RH, Kissinger JC, Ajioka JW, Sibley LD. Recent expansion of Toxoplasma through enhanced oral transmission. Science. 2003;299:414–6. DOIPubMedGoogle Scholar
- Nei M, Li WH. Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci U S A. 1979;76:5269–73. DOIPubMedGoogle Scholar
- Swofford DL. Phylogenetic analysis using parsimony (* and other methods). Vol. 4.0b10. Sunderland (MA): Sinauer Associates; 2002.
- Higgins DG, Thompson JD, Gibson TJ. Using CLUSTAL for multiple sequence alignments. Methods Enzymol. 1996;266:382–402. DOIPubMedGoogle Scholar
- Frenkel JK. Pathophysiology of toxoplasmosis. Parasitol Today. 1988;4:273–8. DOIPubMedGoogle Scholar
- Dupouy-Camet J, de Souza SL, Maslo C, Paugam A, Saimot AG, Benarous R, Detection of Toxoplasma gondii in venous blood from AIDS patients by polymerase chain reaction. J Clin Microbiol. 1993;31:1866–9.PubMedGoogle Scholar
- Tirard V, Niel G, Rosenheim M, Katlama C, Ciceron L, Ogunkolade W, Diagnosis of toxoplasmosis in patients with AIDS by isolation of the parasite from the blood. N Engl J Med. 1991;324:634. DOIPubMedGoogle Scholar
- Li WH, Graur D. Fundamentals of molecular evolution. Sunderland (MA): Sinauer Associates; 1991.
- Neto EC, Anele E, Rubim R, Brites A, Schulte J, Becker D, High prevalence of congenital toxoplasmosis in Brazil estimated in a 3-year prospective neonatal screening study. Int J Epidemiol. 2000;29:941–7. DOIPubMedGoogle Scholar
- Petersen E, Pollak A, Reiter-Owona I. Recent trends in research on congenital toxoplasmosis. Int J Parasitol. 2001;31:115–44. DOIPubMedGoogle Scholar
- Santos CBA, Carvalho ACFB, Ragozo AMA, Soares RM, Amaku M, Yai LEO, First isolation and molecular characterization of Toxoplasma gondii from finishing pigs from São Paulo State, Brazil. Vet Parasitol. 2005;131:207–11. DOIPubMedGoogle Scholar
- Dubey JP, Graham DH, Blackston CR, Lehmann T, Gennari SM, Ragozo AMA, Biological and genetic characterisation of Toxoplasma gondii isolates from chickens (Gallus domesticus) from São Paulo, Brazil: unexpected findings. Int J Parasitol. 2002;32:99–105. DOIPubMedGoogle Scholar
- Silva AV, Pezerico SB, Lima VY, d'Arc Moretti L, Pinheiro JP, Tanaka EM, Genotyping of Toxoplasma gondii strains isolated from dogs with neurological signs. Vet Parasitol. 2005;127:23–7. DOIPubMedGoogle Scholar
- Dubey JP, Navarro IT, Sreekumar C, Dahl E, Freire RL, Kawabata HH, Toxoplasma gondii infections in cats from Paraná, Brazil: seroprevalence, tissue distribution, and biologic and genetic characterization of isolates. J Parasitol. 2004;90:721–6. DOIPubMedGoogle Scholar
- Grigg ME, Ganatra J, Boothroyd JC, Margolis TP. Unusual abundance of atypical strains associated with human ocular toxoplasmosis. J Infect Dis. 2001;184:633–9. DOIPubMedGoogle Scholar
- Howe DK, Honoré S, Derouin F, Sibley LD. Determination of genotypes of Toxoplasma gondii strains isolated from patients with toxoplasmosis. J Clin Microbiol. 1997;35:1411–4.PubMedGoogle Scholar
- Dubey JP, Graham DH, da Silva DS, Lehmann T, Bahia-Oliveira LM. Toxoplasma gondii isolates of free-ranging chickens from Rio de Janeiro, Brazil: mouse mortality, genotype, and oocyst shedding by cats. J Parasitol. 2003;89:851–3. DOIPubMedGoogle Scholar
- Dubey JP, Venturini MC, Venturini L, Piscopo M, Graham DH, Dahl E, Isolation and genotyping of Toxoplasma gondii from free-ranging chickens from Argentina. J Parasitol. 2003;89:1063–4. DOIPubMedGoogle Scholar
- Dubey JP, Gomez-Marin JE, Bedoya A, Lora F, Vianna MC, Hill D, Genetic and biologic characteristics of Toxoplasma gondii isolates in free-range chickens from Colombia, South America. Vet Parasitol. 2005;134:67–72. DOIPubMedGoogle Scholar
- Dubey JP, Karhemere S, Dahl E, Sreekumar C, Diabate A, Dabire KR, First biological and genetic characterization of Toxoplasma gondii isolates from chickens from Africa (Democratic Republic of Congo, Mali, Burkina Faso, and Kenya). J Parasitol. 2005;91:69–72. DOIPubMedGoogle Scholar
- Dubey JP, Graham DH, Dahl E, Hilali M, El-Ghaysh A, Streekumar C, Isolation and molecular characterization of Toxoplasma gondii from chickens and ducks from Egypt. Vet Parasitol. 2003;114:89–95. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 12, Number 6—June 2006
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Address correspondence to: L. David Sibley, Washington University Medical School, 660 S Euclid Ave, Saint Louis, MO 63130, USA
Top