Volume 17, Number 7—July 2011
Dispatch
Age as Risk Factor for Death from Pandemic (H1N1) 2009, Chile
Abstract
Pandemic (H1N1) 2009 affected Chile during the winter of 2009. The hospitalization rate was 0.56% overall and 3.47% for persons >60 years of age at risk for severe disease and death independent of concurrent conditions. Age >60 years was the major risk factor for death from pandemic (H1N1) 2009.
On April 23, 2009, the World Health Organization issued alerts about the emergence of pandemic (H1N1) 2009. On May 17, during fall in the Southern Hemisphere, the first case of this disease in Chile was identified (1). Pandemic (H1N1) 2009 then replaced seasonal influenza, which had accounted for <1% of confirmed influenza cases in Chile (2). Immediately after the alerts, a national mandatory notification system was started in Chile for influenza-like illness (ILI) cases and hospitalized persons with pandemic (H1N1) 2009 in public and private institutions. We analyzed data for patients with confirmed pandemic (H1N1) 2009 hospitalized during epidemiologic weeks 20–32 (May 19–August 3), 2009.
ILI was defined as fever >38.5°C and cough plus >1 of the following: sore throat, headache, and myalgia. Severe influenza was defined as any case confirmed by reverse transcription PCR in a hospitalized person. The National Ministry of Health provided oseltavimir to every patient >5 years of age who had ILI. Approximately 80% of ILI cases corresponded to pandemic (H1N1) 2009 confirmed by PCR in a pilot study.
All fatal cases included in the analysis occurred in persons whose deaths were directly attributable to influenza. Demographic data, clinical features, concurrent conditions, and number of consultations before hospitalization were recorded (3).
Denominators used for determining overall incidence rates and incidence rates by age group were based on the national census of 2002. Denominators used for determining rates for hospitalized case-patients and case-fatality rates (CFRs) were 80% of ILI cases reported to the Ministry of Health during the study. We conducted statistical analysis by using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA) and Epi Info version 6 (Centers for Disease Control and Prevention, Atlanta, GA, USA).
During May 19–August 3, 2009, a total of 342,588 ILI cases were reported. Median age of case-patients was 20.4 years (range <1–109 years). The overall attack rates were 1.2% for pandemic (H1N1) 2009 and 0.4% for persons >60 years of age. During the same period, 651,416 treatments with oseltamivir were reported. Treatment data indicated that the attack rate was 4%.
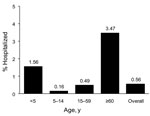
A total of 1,585 persons confirmed to have ILI were hospitalized. Median age was 33 years (range 11 days–94 years), and 52% were women. Overall rate of hospitalization was 9.4/100,000 persons. Case-hospitalization rate was 0.6% (3.5% for persons >60 years of age and 1.6% for children <5 years of age) (Figure 1). Pneumonia was the most common diagnosis at admission (77.3%). Underlying diseases were present in 560 (56.6%) of 989 case-patients, and 217 (22%) had >2 concurrent conditions.
A total of 130 hospitalized patients died (overall mortality rate 0.79/100,000 persons). Among these patients were 117 who died during the study period. Median age was 47 years (range 4 months–89 years) for persons who died and 30 years (range <1–94 years) for persons who survived (p<0.0001). Forty-seven percent of patients who died were >49 years of age. CFR was 0.04%: the highest rate was for patients >60 years of age (0.44%; p<0.0000001), followed by persons 15–59 years of age (0.045%). CFR was 0.02% for children <5 years of age and 0.008% for children 5–14 years of age (Figure 2). Fifty percent of all deaths were caused by severe respiratory failure.
Age was a risk factor for severe influenza and death. Patients >60 years of age and <5 years of age were at a higher risk for severe disease, and patients >60 years of age were at a higher risk for death. At least 1 concurrent condition was identified as a risk factor for death from pandemic (H1N1) 2009. Eighty-four (87.5%) of 96 patients who died and 485 (54.3%) of 893 of patients who survived had an underlying disease (odds ratio 5.89, 95% confidence interval 3.08–11.52; p<0.00001). A concurrent condition was a risk only for persons 15–49 years of age (Table).
Mean time from onset of symptoms to hospitalization was 4.3 days (range 0–20 days) for persons who died and 3.6 days (range 0–20 days) for persons who survived (p = 0.03). Median time from onset of symptoms to beginning of treatment with oseltamivir was 3.0 days for persons who died and for those who survived.
The strength of this study was inclusion of nearly all patients with confirmed severe pandemic (H1N1) 2009 in Chile because of timely implementation of a national notification system. The population studied included 97% of hospitalized patients with pandemic (H1N1) 2009.
Differences in attack rate (1.2% by reporting of ILI cases and 4% by amount of oseltamivir provided) might be explained by the fact that not all ILI cases were captured (≈20% were lost because of underreporting) by the notification system and oseltamivir was prescribed to persons with illness not included in the ILI case definition. We demonstrated in a pilot study that ≈60%–70% of oseltamivir used complied with the case definition for ILI.
The first wave of pandemic (H1N1) 2009 in Chile showed the highest incidence rate in children 5–14 years of age; persons ≥60 years of age were least affected (2). However, our study identified persons ≥60 years of age as at greatest risk for severe respiratory disease and death, despite the lower incidence rates. During the influenza pandemic, risk for illness in this age group was low but risk for severe disease and death was higher than in the other groups, independent of underlying diseases. This finding differs from results of a study in California, USA, in which persons >60 years of age did not have a high hospitalization rate (4). Our finding is consistent with those of a report on seasonal influenza in which risk for severe infection and hospitalization was highest for elderly persons (5).
Before April 2009, influenza was rarely reported as the cause of death in Chile. Therefore, only CFRs for severe respiratory infection are available for comparison. The CFRs for patients >65 years during winter 2009 was similar to that during previous winters. We conclude that elderly persons’ risk for a severe outcome during pandemic (H1N1) 2009 did not exceed this risk for a severe outcome during seasonal influenza. Previous reports have documented increased risk for severe outcomes in younger persons during pandemic (H1N1) 2009 and the absence of increased risk for disease severity among elderly persons (4,6,7). On the basis of these findings, some institutions made policy decisions to exclude anyone >65 years of age without concurrent conditions from satisfying recommendations for use of vaccine against pandemic (H1N1) 2009 because of absence of identified increased risk for infection. Our study indicates an age >60 is the greatest risk factor for a severe outcome during pandemic (H1N1) 2009 and seasonal influenza.
Delay in medical care was another risk factor for death in this study. The number of consultations before admission did not differ between the groups, suggesting that patients who died sought medical care later than patients who survived. Thus, timely medical consultation affected patient outcome.
This study indicates that an age ≥60 years was the greatest risk for death associated with pandemic (H1N1) 2009 influenza, similar to that for seasonal influenza. These results can be used for future planning strategies for influenza, strengthening the need for influenza vaccination, opportune medical evaluation, and timely therapy specific for this age group.
Dr Dabanch is an infectious disease specialist at Hospital Militar, Santiago, Chile, and an assistant professor at Universidad de los Andes, Santiago. Her primary research interest is pandemic influenza.
Acknowledgments
Additional members of the Advisory Committee of the Chilean Ministry of Health who contributed data to this study are Jorge Jimenez, Enrique Paris, and Carlos Pérez (Pontificia Universidad Católica de Chile, Santiago); Miguel O’Ryan (Universidad de Chile, Santiago); and Luis Miguel Noriega and Pablo Vial (Universidad del Desarrollo, Santiago).
We thank Winston Andrade, Patricia Bustos, Graciela Torres, and Judith Mora for helping with influenza diagnosis of all hospitalized patients with suspected influenza during the outbreak, and the physicians for reporting ambulatory and hospitalized patients with influenza.
References
- Ministerio de Salud. Influenza pandémica (H1N1) 2009. Report 30, December 2009 [cited 2011 Jan 14]. www.pandemia.cl/templates/pandemia/documentos/prensa/comunicado_1_caso_inf_ah1n1.pdf
- Ministerio de Salud. Vigilancia de influenza. Boletín electrónico no. 80 [cited 2011 Jan 14]. http://epi.minsal.cl/epi/html/AtlasInteractivos/Atlas_BEM80/Influenza.htm
- Ministerio de Salud. Formulario de notificación inmediata para vigilancia de influenza humana H1N1, IRA grave y envío de muestras a confirmación [cited 2011 Jan 14]. http://www.pandemia.cl/templates/pandemia/documentos/fORMULARIO_NOTIF%20IRAG_2010_nueva_circular1.pdf
- Louie JK, Acosta M, Winter K, Jean C, Gavali S, Schechter R, Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA. 2009;302:1896–902. DOIPubMedGoogle Scholar
- Thompson WW, Shay D, Weintraub E, Brammer L, Bridges C, Cox N, Influenza-associated hospitalizations in the United States. JAMA. 2004;292:1333–40. DOIPubMedGoogle Scholar
- Centers for Diseases Control and Prevention. CDC estimates of 2009 H1N1 influenza cases, hospitalizations and deaths in the United States, April–December 12, 2009 [cited 2011 Jan 14]. http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm
- Hanslik T, Boelle P, Flahault A. Preliminary estimation of risk factors for admission to intensive care units and for death in patients infected with A(H1N1)2009 influenza virus, France, 2009–2010. PLoS Curr. 2010;2:RRN1150. DOIPubMedGoogle Scholar
Figures
Table
Cite This Article1These authors contributed equally to this article.
2Additional members of this committee who contributed data to this study are listed at the end of this article.
Table of Contents – Volume 17, Number 7—July 2011
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Cecilia Perret, Division of Pediatrics, Department of Infectious Diseases, Pontificia Universidad Católica de Chile, Santiago, Chile
Top