Volume 19, Number 4—April 2013
Research
Effect of 10-Valent Pneumococcal Vaccine on Pneumonia among Children, Brazil
Figure 5
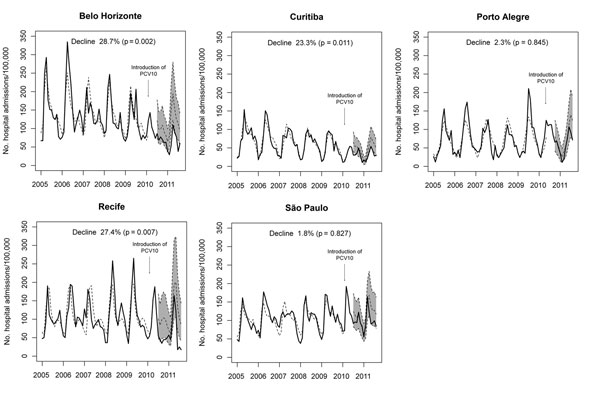
Figure 5. . . Observed (solid lines) and predicted (dashed lines) rates of hospitalization for pneumonia and 95% CIs (shaded area) among children 2 months–2 years of age in 5 cities, Brazil, January 2005–August 2011. The 95% CIs are shown only for the 4 months after start of vaccination. Decline represents the reduction in hospitalizations for pneumonia. PCV10, 10-valent pneumococcal vaccine.
Page created: March 13, 2013
Page updated: March 13, 2013
Page reviewed: March 13, 2013
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.