Volume 25, Number 1—January 2019
Dispatch
Dengue Virus IgM Serotyping by ELISA with Recombinant Mutant Envelope Proteins
Figure 1
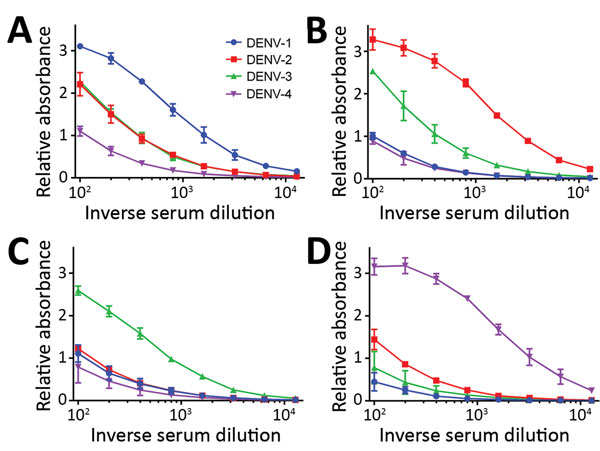
Figure 1. DENV IgM ELISA titers, by serotype, for DENV PCR–positive serum samples from travelers returning to Germany or Italy, 2013–2016. A) DENV-1; B) DENV-2; C) DENV-3; D) DENV-4. Data lines indicate average titers; error bars indicate SDs. The antigens in this ELISA were Equad proteins (i.e., envelope protein from each DENV serotype with 4 amino acid changes T76R, Q77E, W101R, and L107R). In these examples, the highest endpoint titers corresponded to the DENV serotype identified by PCR analysis. DENV, dengue virus.
Page created: December 18, 2018
Page updated: December 18, 2018
Page reviewed: December 18, 2018
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.