Volume 9, Number 10—October 2003
Dispatch
Severe Histoplasmosis in Travelers to Nicaragua
Abstract
We investigated an outbreak of unexpectedly severe histoplasmosis among 14 healthy adventure travelers from the United States who visited a bat-infested cave in Nicaragua. Although histoplasmosis has rarely been reported to cause serious illness among travelers, this outbreak demonstrates that cases may be severe among travelers, even young, healthy persons.
Histoplasmosis is a systemic infection caused by the dimorphic fungus, Histoplasma capsulatum. Infection results from inhaling spores, usually through exposure to bat and bird droppings in barnyards and caves. Although outbreaks have occurred after visits to bat-infested caves, histoplasmosis has not been frequently recognized as travel-related and has rarely led to serious illness among young, healthy travelers. This fungus is endemic in the United States along the Ohio and Mississippi River valleys and many other parts of the world, particularly Latin America. Histoplasmosis is often asymptomatic in endemic settings, but infection can result in a spectrum of illness, ranging from mild influenzalike illness to acute pulmonary infection and disseminated extrapulmonary disease. Immunocompromised persons and the elderly are at greater risk for disseminated disease (1). This report describes a recent outbreak of histoplasmosis among U.S. adventure travelers to Nicaragua that was associated with a high attack rate and hospitalizations among previously healthy travelers.
In June 2001, five persons with a febrile respiratory illness visited a community hospital. The patients were among 15 persons (age range 19–61 years; median 38 years), residents of four states, who had participated in a geology-biology community college class trip to Nicaragua. To determine the cause and describe characteristics of the outbreak, we interviewed trip participants and reviewed medical records to collect demographic information, clinical history, and activities during the trip. Based on the partcipant’s exposure to a cave with bats and clinical characteristics suggestive of acute histoplasmosis, diagnostic testing for histoplasmosis was conducted.
A case of acute histoplasmosis was defined as illness in a person who tested positive by one of the following laboratory tests: serology, urine antigen, histopathology, or culture. Immunodiffusion and complement- fixation serologic tests were performed at the Centers for Disease Control and Prevention (CDC). The immunodiffusion test was considered positive if an H or M band or both were detected. A complement-fixation titer to the yeast or mycelial antigens of >1:32 or a fourfold increase in the titer between acute- and convalescent-phase serum specimens was considered evidence of acute infection. Acute-phase serum specimens were obtained 2–3 weeks after exposure. Convalescent-phase serum specimens were collected approximately 3 months after exposure. One patient underwent bronchoalveolar lavage and bronchoscopy, and a biopsy specimen was obtained. Lung tissue was stained with hematoxylin and eosin and Gomori methenamine silver stains. Bronchoalveolar lavage washings were cultured by conventional methods (2). Identification of the organism as H. capsulatum was confirmed by GenProbe assay (GenProbe, San Diego, CA). Urine antigen tests, which were performed 2 weeks after exposure, are considered positive if >1 unit is detected (3,4).
The travelers began their trip on May 19 and returned to the United States on May 30. During their trip, they explored rock quarries, visited a biological research station, swam in freshwater lakes, and were exposed to parrots and insects. On May 21, a total of 14 of the 15 travelers entered a small cave that had previously been used as a silver mine. They remained in the cave for 10 minutes, during which time they saw several flying bats and bat guano on the ground.
The infection rate was 100% among the 14 travelers who entered the cave, and 12 (86%) were symptomatic. All 14 patients had serologically confirmed infection. Of the 14 patients, 12 (86%) had urine antigen tests (Table). The only traveler who did not enter the cave tested negative by serology and urine antigen.
Of the infected travelers, 8 (57%) were female. The average incubation period was 11 days (range 10–12 days), assuming that exposure occurred on May 21. Among the 12 symptomatic persons, 12 (100%) had a fever of 38.9°C to 40°C, nonproductive cough, myalgias, fever, chills, night sweats, loss of appetite, and mild headache. Many had nausea and vomiting (83%), chest pain (58%), and arthralgias (58%). Six persons had mild to moderate respiratory distress, with oxygen saturation of 88% to 95%. On physical examination, all the travelers’ lung fields were clear to auscultation, a feature consistent with histoplasmosis infection. The two asymptomatic persons had previous exposure to bats or caving. Among the symptomatic persons, we found no differences in severity of symptoms according to sex, prior exposure to spelunking, residence in a histoplamosis-endemic area, or activities while in the mine.
In accordance with clinical treatment guidelines, physicians decided to treat patients with itraconazole for 6 to 12 weeks; some patients also received steroids (5). No patients required discontinuation of therapy was discontinued because of adverse effects.
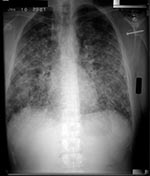
Laboratory studies showed normal complete blood counts, serum chemistries, and renal function tests. Four persons had mild to moderate elevation in liver function tests (alanine aminotransferase, range 55–204; aspartate aminotransferase, range 48–153; and alkaline phosphatase, range 95–311). All 12 symptomatic persons had abnormal chest radiograph results with bilateral nodular infiltrates (Figure). Two persons had hilar adenopathy, and one had a small pleural effusion.
Hematoxylin and eosin–stained sections of the lung tissue from the biopsy showed macrophages and neutrophils in the interstitium; no granulomatous inflammation was observed. Gomori methenamine silver stain demonstrated budding yeasts compatible with H. capsulatum. Culture of the bronchoalveolar lavage fluid from this patient grew H. capsulatum after 4 weeks of incubation.
This report highlights the importance of histoplasmosis as a potentially serious travel-related illness. Histoplasmosis is usually considered a mild, self-limited illness; however, this outbreak of histoplasmosis among previously healthy travelers was associated with a 100% infection rate of histoplasmosis and an 86% rate of symptomatic infection in the persons who entered the cave. Among the symptomatic persons, 50% required hospitalization and 83% missed school or work (range: 2 weeks–3 months) as a result of their illness (Table). Because of the high attack rate and the large percentage of patients with severe infection, these travelers were likely exposed to very high concentrations of H. capsulatum spores during their brief visit inside the cave.
H. capsulatum exists throughout the world (1,6); however, nonimmune travelers from areas with a low prevalence of histoplasmosis, who engage in high-risk activities in disease-endemic areas, are at greater risk of acquiring symptomatic fungal infection. The number of outbreaks of histoplasmosis, especially among U.S. travelers to Latin America, has increased. Histoplasmosis has been reported in groups of travelers who entered caves with bats in Costa Rica (7; unpub. data, Centers for Disease Control and Prevention CDC), Ecuador (8), and Peru (9). In 2001, more than 200 college students became infected with histoplasmosis during a spring break trip to Acapulco, Mexico (10,11). Outbreaks and isolated cases of histoplasmosis have also been described among non–-U.S. travelers to Latin America and other areas of the world (12–19).
Clinicians should consider fungal pathogens when evaluating returning travelers who have a febrile respiratory syndrome. The differential diagnosis of acute febrile respiratory illness in international adventure travelers is extensive and includes legionellosis, psittacosis, leptospirosis, schistosomiasis, histoplasmosis, coccidioidomycosis, influenza, parainfluenza, mycoplasma, dengue, and malaria.
The timely diagnosis of histoplasmosis in travelers may be particularly challenging for clinicians evaluating one sporadic case or a patient involved in an undetected multistate outbreak. Antibody response can take 4–6 weeks to develop, reducing its usefulness in the acute setting and requiring the collection of a convalescent-phase specimen for confirmation. Antibodies may also persist for 5 years, making it difficult to distinguish between prior and recent infection without obtaining a convalescent-phase specimen. The sensitivity of immunodiffusion is 72% to 85%, while that of complement fixation is 80% to 90% (20,21). The antigen test can be used for urine, cerebrospinal fluid, serum, and bronchoalveolar lavage fluid, and results are available in 24 hours. This test had been used most extensively in AIDS patients; however, it is less sensitive in nonimmunocompromised persons with acute pulmonary infection. Sensitivity of the urine antigen test for detection of acute pulmonary infection ranges from 44% to 75% and increases when performed early after the onset of symptoms (3,4).
In this outbreak, 80% of the travelers were prescribed appropriate malaria chemoprophylaxis during a pretravel assessment. However, most clinicians may not have thought to counsel them about histoplasmosis risk and prevention. During pretravel visits, clinicians should review patients’ itineraries for possible fungal exposures and should counsel them to avoid entering caves, especially those known to be bat infested. Since travelers may not know whether their itinerary includes visits to bat-infested caves, adventure travelers and persons who are at high risk for severe histoplasmosis infection, such as immunocompromised travelers (especially persons with AIDS), should be counseled to avoid possible exposures, either by avoiding caves or by using special protective masks. The National Institute for Occupational Safety and Health has published guidelines for special masks that can be used to reduce occupational and other exposures (e.g., among spelunkers). Unfortunately, these special masks are bulky and may be impractical for many travelers (22). While most nonimmunocompromised patients with acute pulmonary histoplasmosis improve without treatment, persons with hypoxemia, diffuse pulmonary histoplasmosis, or severe illness requiring hospitalization may benefit from antifungal therapy and, in some cases, corticosteroids (5).
The contribution of histoplasmosis to infectious disease illness in travelers is likely underestimated. As adventure travel becomes increasingly accessible, especially among persons at high risk, immunocompromised persons, and the elderly, histoplasmosis and other fungal infections in travelers may become more common. Enhanced surveillance for histoplasmosis and other fungal infections by public health officials, combined with heightened awareness by clinicians who are evaluating symptomatic, post-travel patients, can lead to greater understanding of the epidemiology of fungal infections among travelers and, ultimately, to improved prevention measures.
Dr. Weinberg is the team leader for Migrant and Border Health in the Surveillance and Epidemiology Branch, Division of Global Migration and Quarantine, Centers for Disease Control and Prevention. Her research interests include international health, particularly issues in Hispanic populations. She has coordinated the binational activities of the U.S–- Mexico Border Infectious Diseases Surveillance network since 1999.
Acknowledgments
We thank Juan Jose Amador of Nicaraguan Ministry of Health, Phyllis Kozarsky, the members of the group who participated in this trip, and the Histoplasmosis Reference Laboratory, which performed the urine antigen testing, for their help with this investigation, and Ava Navin for editorial assistance.
When the work was done for this outbreak investigation, Ms. Connolly was associated with Indiana University. She is currently an employee of MiraVista Diagnostics, Indianapolis, Indiana.
References
- Deepe G. Histoplasma capsulatum. In: Mandel GL, Bennett JE, Dolin R, editors. Principles and practice of infectious diseases. 5th ed. New York: Churchill Livingston; 2000. p. 2718–33.
- Larone DH, Mitchell TG, Walsh TJ. Histoplasma, blastomyces, coccidioides, and other dimorphic fungi causing systemic mycoses. In: Murray PR, editor. Manual of clinical microbiology. 7th ed. Washington: ASM Press; 1999. p. 1259–68.
- Williams B, Fojtasek M, Connolly-Stringfield P, Wheat J. Diagnosis of histoplasmosis by antigen detection during an outbreak in Indianapolis, Ind. Arch Pathol Lab Med. 1994;118:1205–8.PubMedGoogle Scholar
- Wheat LJ, Kohler RB, Tewari RP. Diagnosis of disseminated histoplasmosis by detection of Histoplasma capsulatum antigen in serum and urine specimens. N Engl J Med. 1986;314:83–8. DOIPubMedGoogle Scholar
- Wheat J, Sarosi G, McKinsey D, Hamill R, Bradsher R, Johnson P, Practice guidelines for the management of patients with histoplasmosis. Clin Infect Dis. 2000;30:688–95. DOIPubMedGoogle Scholar
- Wilson ME. A world guide to infections: diseases, distribution, diagnosis. Oxford: Oxford University Press; 1991. p.190.
- Centers for Disease Control and Prevention. Cave-associated histoplasmosis—Costa Rica. MMWR Morb Mortal Wkly Rep. 1988;37:312–3.PubMedGoogle Scholar
- Valdez H, Salata RA. Bat-associated histoplasmosis in returning travelers: case presentation and description of a cluster. J Travel Med. 1999;6:258–60. DOIPubMedGoogle Scholar
- Nasta P, Donisi A, Cattane A, Chiodera A, Cassari S. Acute histoplasmosis in spelunkers returning from Mato Grosso, Peru. J Travel Med. 1997;4:176–8. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Outbreak of acute respiratory febrile illness among college students—Acapulco, Mexico, March 2001. MMWR Morb Mortal Wkly Rep. 2001;50:261–2.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: outbreak of acute febrile respiratory illness among college students—Acapulco, Mexico, March 2001. MMWR Morb Mortal Wkly Rep. 2001;50:359–60.PubMedGoogle Scholar
- Bonnet D, Balandraud P, Lonjon T, Rey P, Van de Walle JP, Cador L, Round pulmonary lesions after returning from French Guyana. Six cases of American pulmonary histoplasmosis. Med Trop. 1995;55:55–60.PubMedGoogle Scholar
- Buxton JA, Dawar M, Wheat LJ, Black WA, Ames NG, Mugford M, Outbreak of histoplasmosis in a school party that visited a cave in Belize: role of antigen testing in diagnosis. J Travel Med. 2002;9:48–50. DOIPubMedGoogle Scholar
- Erkens K, Lademann M, Tintelnot K, Lafrenz M, Kaben U, Reisinger EC. Histoplasmosis group disease in bat researchers returning from Cuba. Dtsch Med Wochenschr. 2002;127:21–5. DOIPubMedGoogle Scholar
- Fujio J, Nishimura K, Miyaji M. Epidemiological survey of the imported mycoses in Japan. Nippon Ishinkin Gakkai Zasshi. 1999;40:103–9. DOIPubMedGoogle Scholar
- Gascon J, Torres JM, Luburich P, Ayuso JR, Xaubet A, Corachan M. Imported histoplasmosis in Spain. J Travel Med. 2000;7:89–91. DOIPubMedGoogle Scholar
- Hatakeyama S, Kashiyama T, Takechi A, Sasaki S, Akamatsu E. Cave-associated acute pulmonary histoplasmosis in two Japanese returning from Mexico. Nihon Kokyuki Gakkai Zasshi. 2001;39:293–7.PubMedGoogle Scholar
- Hirsch D, Leupold W, Rupprecht E. Pulmonary histoplasmoma after travel abroad. Pneumologie. 1996;50:242–4.PubMedGoogle Scholar
- Suzaki A, Kimura M, Kimura S, Shimada K, Miyaji M, Kaufman L. An outbreak of acute pulmonary histoplasmosis among travelers to a bat-inhabited cave in Brazil. Kansenshogaku Zasshi. 1995;69:444–9.PubMedGoogle Scholar
- Morrison CJ, Lindsley MD. Serological approaches to the diagnosis of invasive fungal infections. In: Calderone R, Cihlar R, editors. Fungal pathogenesis: principles and practice. New York: Marcel Dekker; 2001. p. 667–716.
- Reiss E, Kaufman L, Kovacs JA, Lindsley MD. Clinical immunomycology. In: Rose NR, Hamilton RG, Detrick B, editors. Manual of clinical laboratory immunology. Sixth edition. Washington: American Society for Microbiology; 2002. p. 559–83.
- Centers for Disease Control and Prevention. National Institute for Occupational Safety and Health. Histoplasmosis: protecting workers at risk, revised guidelines for preventing histoplasmosis; 1997. Available from: URL: http://www.cdc.gov/niosh/97-146.html
Figure
Table
Cite This ArticleTable of Contents – Volume 9, Number 10—October 2003
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Michelle Weinberg, Division of Global Migration and Quarantine, Centers for Disease Control and Prevention, 1600 Clifton Rd, Mailstop E03, Atlanta, Georgia, USA, 30333; fax: 404-498-1633
Top