Volume 20, Number 10—October 2014
Research
Effects of Mefloquine Use on Plasmodium vivax Multidrug Resistance
Abstract
Numerous studies have indicated a strong association between amplification of the multidrug resistance-1 gene and in vivo and in vitro mefloquine resistance of Plasmodium falciparum. Although falciparum infection usually is not treated with mefloquine, incorrect diagnosis, high frequency of undetected mixed infections, or relapses of P. vivax infection triggered by P. falciparum infections expose non–P. falciparum parasites to mefloquine. To assess the consequences of such unintentional treatments on P. vivax, we studied variations in number of Pvmdr-1 (PlasmoDB accession no. PVX_080100, NCBI reference sequence NC_009915.1) copies worldwide in 607 samples collected in areas with different histories of mefloquine use from residents and from travelers returning to France. Number of Pvmdr-1 copies correlated with drug use history. Treatment against P. falciparum exerts substantial collateral pressure against sympatric P. vivax, jeopardizing future use of mefloquine against P. vivax. A drug policy is needed that takes into consideration all co-endemic species of malaria parasites.
Since World War II, antimalarial drugs have been intensively used to prevent or treat malaria (1). As observed with other antimicrobial agents, their use (or frequent misuse, when malaria diagnosis was based only on clinical symptoms without parasitologic confirmation) led to the emergence, selection, and spread of resistant parasites (2). This resistance became a global problem during the 1960s, when Plasmodium falciparum parasites developed resistance to chloroquine, the most widely used antimalarial drug at that time (3). In particular, resistant parasites that emerged in the Greater Mekong subregion of Asia later spread to Africa, triggering a dramatic increase in malaria and malaria-related deaths, particularly among children (4). During the 1980s, a similar scenario was observed with sulfadoxine-pyrimethamine (SP) when this association was recommended to replace chloroquine as first-line treatment in uncomplicated falciparum malaria (5,6). Since then, biological and molecular investigations using laboratory and field isolates have demonstrated that resistance of P. falciparum to antimalarial drugs is mediated by 2 major mechanisms: 1) a modification of the parasite target (i.e., mutations in dihydrofolate reductase [dhfr] or in dihydropteroate synthetase [dhps] genes) or 2) an increase of the efflux of the drug away from its site of action (i.e., mutations in the chloroquine resistant transporter gene or the multidrug resistance-1 [mdr-1] gene or in an increased number of copies of the mdr-1 gene). These molecular events have been intensively studied and are well known for P. falciparum but not for other Plasmodium species, mainly because of the ability to culture in vitro P. falciparum erythrocytic stages.
Our understanding of the molecular mechanisms of antimalarial drug resistance developed by P. vivax is less comprehensive. Although Aotus and Saimiri monkey models have provided useful information about P. vivax biology, most of the data have been gained through comparative studies investigating polymorphisms in orthologous genes encoding resistance to pyrimethamine (dhfr gene), sulfadoxine (dhps gene), or chloroquine (chloroquine resistant transporter or mdr-1 genes). For instance, mutations in codons 57, 58, 61, 117, and 173 of P. vivax DHFR (corresponding to codons 51, 59, 108, and 164 of P. falciparum DHFR) are involved in resistance to pyrimethamine, although P. vivax infections are not usually treated directly with SP (7). This resistance was confirmed by heterologous expression studies, invalidating the common idea that P. vivax was “intrinsically resistant” to pyrimethamine (8), which suggests that the high frequency of mixed P. falciparum/P. vivax infections that are not detected by microscopy (9–11) or relapses of P. vivax infection after P. falciparum infections probably exposes P. vivax parasites to antimalarial drugs used to treat falciparum malaria infections, especially those with a long half-life, and selects P. vivax genetic traits conferring antimalarial drug resistance.
The impact of antimalarial drugs, especially those with long half-lives (such as mefloquine), on the sympatric Plasmodium species is not clearly understood. In areas where P. falciparum and P. vivax are co-endemic, such as South America and Southeast Asia, mefloquine has been widely used (alone in monotherapy or in combination with artemisinin derivatives) to treat uncomplicated falciparum malaria (12). In both areas, emergence of P. falciparum parasites resistant to mefloquine has been demonstrated from therapeutic efficacy studies (treatment failure) or in vitro testing (increased IC50 [half maximal inhibitory concentration]) and has been associated with the amplification of P. falciparum mdr-1 (Pfmdr-1) gene (13–16). Recently, several studies performed on P. vivax samples collected in Southeast Asia (Thailand, Laos, Cambodia, and Myanmar) (17–19), South America (Brazil, Honduras) (20,21), and Africa (Mauritania) (22) have shown that mdr-1 amplification does occur in P. vivax.
In this context, and to confirm the impact of the mefloquine drug pressure on P. vivax parasite populations, we used a real-time PCR to assess the number of P. vivax mdr-1 (Pvmdr-1) gene copies to evaluate the worldwide distribution of Pvmdr-1 amplification in samples collected from travelers with vivax malaria returning to France and from residents in areas with different histories of mefloquine use (French Guiana, Cambodia, Madagascar, and Sudan).
Sample Collection
P. vivax and P. falciparum samples from Madagascar were collected during 2006–2007 as part of the antimalarial drug resistance network, from symptomatic patients before treatment in 19 health centers located in areas of Madagascar with different epidemiologic patterns of malaria transmission: northern (Antsiranana, Antsohihy, Andapa), western (Mahajunga, Miandrivazo, Maevatanana, Morondava, Tsiroanomandidy, Ampasimpotsy), central (Saharevo, Moramanga), southern (Ihosy, Ejeda, Tolagnaro, Iakora, Ranostara, Toliara), and eastern (Farafangana, Toamasina). In Cambodia, P. vivax and P. falciparum isolates were obtained from symptomatic persons during 2010 in Pailin and Kratie Provinces. Other P. vivax samples were collected 1) from malaria-infected travelers returning to France after visiting Africa (Côte d’Ivoire, Ethiopia, Madagascar, and Mauritania), South America (Bolivia, Brazil, Colombia, French Guiana, Venezuela), and Asia (Bangladesh, Cambodia, India, Indonesia, Laos, Malaysia, Nepal, Pakistan, Sri Lanka) during 1997–2009 and were provided by the National Reference Center for Malaria (Paris, France); and 2) from symptomatic P. vivax–infected persons in French Guiana (2000–2003) or Sudan (2007).
DNA Extraction and PCR Detection of P. falciparum and P. vivax
We extracted parasite DNA from blood spots with Instagene Matrix (BioRad, Marnes-la-Coquette, France) or from whole blood samples using the phenol-chloroform method (23) or the QIAamp DNA Blood Mini Kit (QIAGEN, Courtaboeuf, France), according to the manufacturer’s instructions. Molecular detection and identification of Plasmodium parasites were performed by using real-time PCR as described by Chou et al. (24).
Determination of the Number of Pfmdr-1 Copies in Isolates from Cambodia and Malagasy
We measured number of Pfmdr-1 copies using CFX96 real-time PCR (BioRad) relative to the single copy of the β-tubulin (used as a reference gene). Briefly, PCRs were conducted in 25-μL volumes in a 96-well plate containing 1X HOT FIREPol EvaGreen qPCR Mix Plus (Solis BioDyne, Tartu, Estonia), 0.3 μM of each forward and reverse primer (Pfmdr-1, 5′-TGCATCTATAAAACGATCAGACAAA-3′ and 5′-TCGTGTGTTCCATGTGACTGT-3′; β-tubulin, 5′-TGATGTGCGCAAGTGATCC-3′ and 5′-TCCTTTGTGGACATTCTTCCTC-3′), and 4 μL of template DNA. Amplifications were performed under the following conditions: 94°C for 15 min, followed by 40 cycles of 94°C for 15 s, 58°C for 20 s, and 72°C for 20 s. The number of Pfmdr-1 copies of each sample was measured in triplicate relative to a standard curve by using 4 standards of mixed plasmids cloned into TOPO cloning vector (Invitrogen, Saint Aubin, France): standard 1 (1:1 ratio of Pfmdr-1 and β-tubulin), standard 2 (2:1 ratio of Pfmdr-1 and β-tubulin), standard 3 (3:1 ratio of Pfmdr-1 and β-tubulin) and standard 4 (4:1 ratio of Pfmdr-1 and β-tubulin) and 2 parasite clonal lines used as controls, the 3D7 Africa line (1 copy of Pfmdr-1) and line Dd2 (3 copies of Pfmdr-1), by the ∆CT method (where CT is the cycle threshold). We defined >1.6 copies as a duplication of the gene.
Evaluation of the Number of Pvmdr-1 Copies
We measured number of Pvmdr-1 (Pfmdr-1) copies following the same procedure, relative to the single copy of the β-tubulin. Briefly, PCRs were conducted in 25-μL volumes in a 96-well plate containing 1X HOT FIREPol EvaGreen qPCR Mix Plus (Solis BioDyne), 0.3 μM of each forward and reverse primer (Pvmdr-1, 5′-GCAACTCCATAAAGAACAACATCA-3′ and 5′-TTTGAGAAGAAAAACCATCTTCG-3′; β-tubulin, 5′-CATGTTCGTTAAGATTTCCTGGT-3′ and 5′-GTTAGTGGTGCAAAACCAATCA-3′), and 4 μL of template DNA. Amplifications were performed under the following conditions: 94°C for 15 min, followed by 45 cycles of 94°C for 15 s, 59°C for 30 s, and 72°C for 30 s. The number of Pvmdr-1 copies of each sample was measured in triplicate relative to a standard curve by using 6 standards of mixed plasmids cloned into TOPO cloning as described for Pfmdr-1 (from the standard-1, 1:1 ratio of Pvmdr-1 and β-tubulin to the standard-6, 6:1 ratio of Pvmdr-1 and β-tubulin) by the ∆CT method (Technical Appendix, Figure 1). We defined >1.6 copies as a duplication of the gene.
Statistical Analysis
Microsoft Excel 2010 (Microsoft, Redmond, WA, USA) and MedCalc software (v9.1.0.1, Mariakerke, Belgium) were used for data analysis. Categorical variables were compared by χ2 test, and continuous variables were compared by using the 1-way analysis of variance or Mann-Whitney U test. We considered p values <0.05 as significant.
Ethical Approval
We obtained ethics clearance for the samples used in this study from National Ethics Committee in Cambodia (Ministry of Health), in Madagascar (Ministry of Health), in Sudan (Ministry of Health), and in France (National Reference Center for Malaria). All patients or their parents/guardians provided informed written consent.
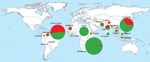
Global Distribution of the Number of Pvmdr-1 Copies
We collected and analyzed 607 P. vivax isolates from areas to which the parasite was endemic from a total of 492 residents (117 in South America;, 117 in Asia; and 258 in Africa) and 115 travelers from France to South America (41 travelers), Asia (60), or Africa (14). The number of Pvmdr-1 copies ranged from 1 to 5 copies (mean 1.28, 95% CI 1.22–1.34; median 1.05 [interquartile range (IQR) 0.84–1.53]) and was distributed as follows: 75% isolates had 1 copy, 18% had 2 copies, 6% had 3 copies, 1.6% had 4 copies, and 0.4% had 5 copies. The frequency of Pvmdr-1–amplified isolates was significantly higher in samples from South America (83 [53%] of 158) than in samples from Asia (60 [34%] of 177, p = 10−3) or Africa (11 [4.0%] of 272, p<10−5). The mean number of Pvmdr-1 copies was also higher in isolates from South America (1.8) than in isolates from Asia (1.3, p = 0.0007) or Africa (0.9, p<10−5). Number of copies differed significantly between P. vivax isolates from residents and those from travelers. In South America, the proportion of isolates with >1 copy of Pvmdr-1 was significantly lower in travelers (34% vs. 59%, odds ratio [OR] 0.4 [95% CI 0.2–0.8], p = 0.007); in Africa, this proportion was significantly higher in travelers (57% vs. 1%, OR 113, 95% CI 24–536, p<0.0001) (Tables 1, 2; Figure 1).
Amplification of Pvmdr-1 in Isolates from Residents
Number of copies in isolates from residents differed significantly among continents (Table 3). The mean number of Pvmdr-1 copies was highest in isolates from South America (2.04, p<10−5 vs. Asia and Africa), followed by Asia (1.32, p<10−4 vs. Africa) and Africa (0.90 for Madagascar and 1.0 for Sudan). In South America, the proportion of isolates with >1 Pvmdr-1 copies was also more frequent (59%) than in Asia (33%, OR 0.33, 95% CI 0.2–0.6, p = 10−4) and Africa (1%, OR 0.008, 95% CI 0.002–0.02, p<10−5 vs. South America; OR = 0.02, 95% CI 0.007–0.08, p<10−4 vs. Asia).
Amplification of Pvmdr-1 in Isolates from Travelers
The mean number of Pvmdr-1 copies was similar for travelers returning to France from South America or Asia and did not change by year of collection: 1997–2000 (1.42 and 1.24, respectively), 2001–2005 (1.17 and 1.38), and 2006–2009 (1.20 and 1.49). In contrast, for travelers returning to France from Africa, the mean number of Pvmdr-1 copies increased in samples collected more recently: 0.60 during 1997–2000, 1.02 during 2001–2005, and 1.48 during 2006–2009 (OR 0.15, 95% CI 0.03–0.71, p = 0.049). The proportion of isolates with >1 copies of Pvmdr-1 did not differ significantly among continents over time (Table 4).
Amplification of Pvmdr-1 and Pfmdr-1 in Isolates from Cambodia and Malagasy
We compared the distribution profile of the number of mdr-1 copies of P. falciparum and P. vivax isolates in 2 different settings. In both Cambodia and Madagascar, the number of mdr-1 copies did not differ between the 2 species (Figure 2). In Cambodia, where mefloquine has been widely used for >25 years, the mean of number of mdr-1 copies was 1.34 for P. falciparum (n = 88, 95% CI 1.22–1.47, range 1–3) and 1.34 for P. vivax (n = 129, 95% CI 1.24–1.44, range 1–4, p = 0.52). In contrast, in Madagascar where mefloquine has never been recommended and has been barely used, the mean number of mdr-1 copies was 0.92 for P. falciparum (n = 350, 95% CI 0.90–0.94, range 1–2) and 0.90 for P. vivax (n = 201, 95% CI 0.87–0.93, range 1–2, p = 0.21).
Developed in the 1970s at the US Department of Defense’s Walter Reed Army Institute of Research as a synthetic analog of quinine (25), mefloquine was introduced in 1983 in Thailand to replace chloroquine as first-line treatment for falciparum malaria (26). Since then, mefloquine alone or in combination with artesunate has been widely used, especially in Southeast Asia (including Cambodia) and South America (including French Guiana), where it was introduced for second-line treatment and for chemoprophylaxis in 1990 (27). In contrast, mefloquine has not been used extensively in Africa and has not been introduced in Madagascar. Mefloquine has been available for malaria chemoprophylaxis since 1985 in Europe and since 1990 in the United States and has been used by >35 million travelers from France for this indication (28,29).
Pfmdr-1 gene amplification has been described as the major mechanism of P. falciparum mefloquine resistance associated with treatment failure or in vitro resistance (13–16). Previous studies, including ours, confirm that mdr-1 amplification does occur in P. vivax (17–22). In addition, the epidemiologic data in our current study show that in regions where mefloquine has never been used, such as in Madagascar and Sudan, amplification in Pvmdr-1 is rare (1% and 2% of total isolates, respectively), whereas in areas with current or past intense use of mefloquine, such as in French Guiana and Cambodia, Pvmdr-1 amplification is frequent and detected in 59% and 33% of isolates, respectively, and with a mean of 2 and 1.3 copies, respectively.
Both the number of copies and prevalence of Pvmdr-1 of isolates with multiple mdr-1 copies we observed here are much higher than reported in other studies. For instance, Imwong et al. reported Pvmdr-1 amplification in 6/66, 2/50, and 1/49 isolates from Thailand, Laos, and Myanmar, respectively (17); Jovel et al. observed Pvmdr-1 amplification in 1/37 in Honduras (20); and Lin et al. recently reported 39% and only 4% prevalence in P. vivax isolates from Thailand and Cambodia, respectively (18). A reason for the discrepancy between observations from Lin et al. in Cambodia and our observations could be because their isolates were collected during 2006–2007 and our samples were collected in 2010, indicating an increase in Pvmdr-1 amplification over 3 years. Another reason is the location of collection. Indeed, drug resistance and drug pressure markedly differ across Cambodia. Lin et al. studied isolates from southern Cambodia (Kampot Province), whereas we studied isolates from areas in which drugs were highly resistant in western (Pailin Province) and southeastern Cambodia (Kratie Province), where multidrug resistance of P falciparum is emerging (30) and drug pressure (including artesunate–mefloquine combination) has been intense in recent years. To our knowledge, the previous maximum number of Pvmdr-1 copies detected was 3 (18,19); in this study. however, we observed up to 5 copies in isolates from South America and 4 copies in isolates from Southeast Asia. This difference is likely to be due to our real-time PCR approach using 6 standards of mixed plasmids, which enabled detection of P. vivax isolates with mdr-1 amplification with up to 6 copies. This observation also could indicate an ongoing selection of mdr-1–amplified parasites. Although, our data need to be confirmed and supported by in vivo data from mefloquine-treated patients or in vitro experiments showing a direct relationship between mefloquine pressure and P. vivax mdr-1 amplification, our findings advocate for an integrated drug policy whereby all sympatric malaria species are considered regarding treatment efficacy but also drug pressure and selection of resistance.
We were able to assess the number of Pvmdr-1 copies in isolates collected from travelers returning to France. These data must be analyzed with caution because information about the location of infection might be erroneous, particularly given the fact that relapses from hypnozoites can occur several months after primary infection. We cannot exclude that a patient declaring having returned from Africa was previously infected during a trip to a different location because we did not include this information in the questionnaire administered at recruitment. Nevertheless, we found significant differences between travelers from France and residents from a given geographic origin, especially in isolates from Africa, where most (99%) isolates from residents displayed no amplification, whereas most (57%) isolates from travelers from France had Pvmdr-1 amplification. We assume this indicates within-host selection by mefloquine prophylaxis, which has been and continues to be widely used among travelers from France who go to malaria-endemic countries. Such pressure does not exist in residents, who usually do not take any prophylaxis. These data are of concern because they suggest that selection of Pvmdr-1 amplification is a more rapid process than previously thought, reminiscent to atovaquone resistance. Although selection in travelers from France returning to nonendemic areas bears no transmission risk, chemoprophylaxis and intermittent preventive treatments in malaria-endemic areas might contribute to the emergence of resistant parasites. This possibility certainly warrants further investigation.
Ms Khim is a PhD student at the Malaria Molecular Epidemiology Unit in Institut Pasteur in Cambodia. Her research interests include clinical epidemiology and antimalarial drug resistance in vivax malaria and more generally are focused on developing molecular tools for improving the surveillance of resistance to antimalarial drugs in Cambodia.
Acknowledgments
We thank all the patients and healthcare workers involved in this study and the staff of the Ministries of Health of Madagascar and Cambodia for their collaboration.
Sample collections and field laboratory work were supported in Madagascar by the Global Fund project for Madagascar round 3 (Community Action to Roll Back Malaria grant no. MDG-304-G05-M) and a Natixis Banques Grant; in Cambodia by the Global Fund Grant Malaria Programme Round 9 (CAM-S10-G14-M); in French Guiana and from French travelers by the French Ministry of Health (InVS agency, Paris); and in Sudan by a grant from the World Health Organization, Global Malaria Programme (HQ/07/100294). Additional funding was provided by the French Ministry of Foreign Affairs (D.M.), the Fondation Pierre Ledoux–Jeunesse Internationale (C.B.), and the Genomics Platform, Pasteur Génopôle, Pasteur Institute (France).
References
- Meshnick S, Dobson M. The history of antimalarial drugs. Totowa (NJ): Humana Press; 2001.
- Wongsrichanalai C, Pickard AL, Wernsdorfer WH, Meshnick SR. Epidemiology of drug-resistant malaria. Lancet Infect Dis. 2002;2:209–18 . DOIPubMedGoogle Scholar
- Wernsdorfer WH, Payne D. The dynamics of drug resistance in Plasmodium falciparum. Pharmacol Ther. 1991;50:95–121. DOIPubMedGoogle Scholar
- Trape JF. The public health impact of chloroquine resistance in Africa. Am J Trop Med Hyg. 2001;64:12–7 .PubMedGoogle Scholar
- Hurwitz ES, Johnson D, Campbell CC. Resistance of Plasmodium falciparum malaria to sulfadoxine-pyrimethamine ('Fansidar') in a refugee camp in Thailand. Lancet. 1981;1:1068–70. DOIPubMedGoogle Scholar
- Mita T, Tanabe K, Kita K. Spread and evolution of Plasmodium falciparum drug resistance. Parasitol Int. 2009;58:201–9. DOIPubMedGoogle Scholar
- Hawkins VN, Joshi H, Rungsihirunrat K, Na-Bangchang K, Sibley CH. Antifolates can have a role in the treatment of Plasmodium vivax. Trends Parasitol. 2007;23:213–22. DOIPubMedGoogle Scholar
- Young MD, Burgess RW. Pyrimethamine resistance in Plasmodium vivax malaria. Bull World Health Organ. 1959;20:27–36 .PubMedGoogle Scholar
- Mayxay M, Pukritrayakamee S, Chotivanich K, Imwong M, Looareesuwan S, White NJ. Identification of cryptic coinfection with Plasmodium falciparum in patients presenting with vivax malaria. Am J Trop Med Hyg. 2001;65:588–92 .PubMedGoogle Scholar
- Mayxay M, Pukrittayakamee S, Newton PN, White NJ. Mixed-species malaria infections in humans. Trends Parasitol. 2004;20:233–40. DOIPubMedGoogle Scholar
- Snounou G, White NJ. The co-existence of Plasmodium: sidelights from falciparum and vivax malaria in Thailand. Trends Parasitol. 2004;20:333–9. DOIPubMedGoogle Scholar
- World Health Organization. World malaria report 2012 [cited 25 Jul 2014]. http://www.who.int/malaria/publications/world_malaria_report_2012/en/
- Cowman AF, Galatis D, Thompson JK. Selection for mefloquine resistance in Plasmodium falciparum is linked to amplification of the pfmdr1 gene and cross-resistance to halofantrine and quinine. Proc Natl Acad Sci U S A. 1994;91:1143–7. DOIPubMedGoogle Scholar
- Peel SA, Bright P, Yount B, Handy J, Baric RS. A strong association between mefloquine and halofantrine resistance and amplification, overexpression, and mutation in the P-glycoprotein gene homolog (pfmdr) of Plasmodium falciparum in vitro. Am J Trop Med Hyg. 1994;51:648–58 .PubMedGoogle Scholar
- Price RN, Cassar C, Brockman A, Duraisingh M, van Vugt M, White NJ, The pfmdr1 gene is associated with a multidrug-resistant phenotype in Plasmodium falciparum from the western border of Thailand. Antimicrob Agents Chemother. 1999;43:2943–9 .PubMedGoogle Scholar
- Price RN, Uhlemann AC, Brockman A, McGready R, Ashley E, Phaipun L, Mefloquine resistance in Plasmodium falciparum and increased pfmdr1 gene copy number. Lancet. 2004;364:438–47. DOIPubMedGoogle Scholar
- Imwong M, Pukrittayakamee S, Pongtavornpinyo W, Nakeesathit S, Nair S, Newton P, Gene amplification of the multidrug resistance 1 gene of Plasmodium vivax isolates from Thailand, Laos, and Myanmar. Antimicrob Agents Chemother. 2008;52:2657–9. DOIPubMedGoogle Scholar
- Lin JT, Patel JC, Kharabora O, Sattabongkot J, Muth S, Ubalee R, Plasmodium vivax isolates from Cambodia and Thailand show high genetic complexity and distinct patterns of P. vivax multidrug resistance gene 1 (pvmdr1) polymorphisms. Am J Trop Med Hyg. 2013;88:1116–23. DOIPubMedGoogle Scholar
- Suwanarusk R, Chavchich M, Russell B, Jaidee A, Chalfein F, Barends M, Amplification of pvmdr1 associated with multidrug-resistant Plasmodium vivax. J Infect Dis. 2008;198:1558–64. DOIPubMedGoogle Scholar
- Jovel IT, Mejia RE, Banegas E, Piedade R, Alger J, Fontecha G, Drug resistance associated genetic polymorphisms in Plasmodium falciparum and Plasmodium vivax collected in Honduras, Central America. Malar J. 2011;10:376. DOIPubMedGoogle Scholar
- Vargas-Rodríguez Rdel C, da Silva Bastos M, Menezes MJ, Orjuela-Sánchez P, Ferreira MU. Single-nucleotide polymorphism and copy number variation of the multidrug resistance-1 locus of Plasmodium vivax: local and global patterns. Am J Trop Med Hyg. 2012;87:813–21 and. DOIPubMedGoogle Scholar
- Mint Lekweiry K, Ould Mohamed Salem Boukhary A, Gaillard T, Wurtz N, Bogreau H, Hafid JE, Molecular surveillance of drug-resistant Plasmodium vivax using pvdhfr, pvdhps and pvmdr1 markers in Nouakchott, Mauritania. J Antimicrob Chemother. 2012;67:367–74. DOIPubMedGoogle Scholar
- Rakotonirina H, Barnadas C, Raherijafy R, Andrianantenaina H, Ratsimbasoa A, Randrianasolo L, Accuracy and reliability of malaria diagnostic techniques for guiding febrile outpatient treatment in malaria-endemic countries. Am J Trop Med Hyg. 2008;78:217–21 .PubMedGoogle Scholar
- Chou M, Kim S, Khim N, Chy S, Sum S, Dourng D, Performance of “VIKIA Malaria Ag Pf/Pan” (IMACCESS®), a new malaria rapid diagnostic test for detection of symptomatic malaria infections. Malar J. 2012;11:295. DOIPubMedGoogle Scholar
- White NJ. Antimalarial drug resistance: the pace quickens. J Antimicrob Chemother. 1992;30:571–85. DOIPubMedGoogle Scholar
- Wongsrichanalai C, Meshnick SR. Declining artesunate-mefloquine efficacy against falciparum malaria on the Cambodia–Thailand border. Emerg Infect Dis. 2008;14:716–9. DOIPubMedGoogle Scholar
- Legrand E, Yrinesi J, Ekala MT, Péneau J, Volney B, Berger F, Discordant temporal evolution of Pfcrt and Pfmdr1 genotypes and Plasmodium falciparum in vitro drug susceptibility to 4-aminoquinolines after drug policy change in French Guiana. Antimicrob Agents Chemother. 2012;56:1382–9. DOIPubMedGoogle Scholar
- Heimgartner E. Practical experience with mefloquine as an antimalarial [in German]. Schweiz Rundsch Med Prax. 1986;75:459–62 .PubMedGoogle Scholar
- World Health Organization. International travel and health. Vaccination requirements and health advice. WHO Office of Information 1995 [cited 2014 Jul 30]. http://www.who.int/ith/en/
- Ariey F, Witkowski B, Amaratunga C, Beghain J, Langlois AC, Khim N, A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature. 2014;505:50–5. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 20, Number 10—October 2014
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Didier Ménard, Malaria Molecular Epidemiology Unit, Institut Pasteur in Cambodia, Phnom Penh, Cambodia
Top