Volume 22, Number 7—July 2016
CME ACTIVITY - Synopsis
Current Guidelines, Common Clinical Pitfalls, and Future Directions for Laboratory Diagnosis of Lyme Disease, United States
Figure 1
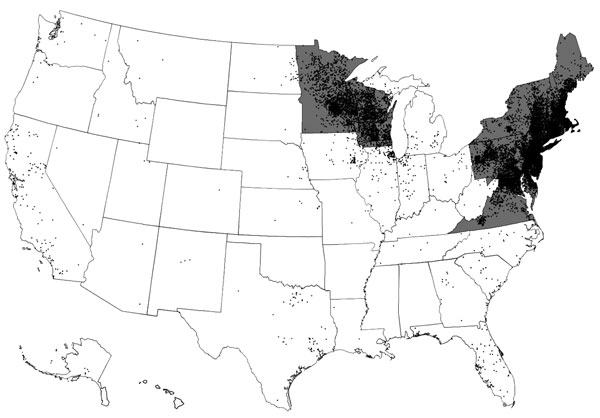
Figure 1. Lyme disease cases (black dots) reported by surveillance, United States, 2005–2010. One dot is placed randomly within the county of residence for each confirmed case. States with the highest incidence of clinician-diagnosed Lyme disease in a large health insurance claims database (gray areas) are also shown. Transmission also occurs in small regions of northern California, Oregon, and Washington. Adapted from (4).
References
- Stanek G, Wormser GP, Gray J, Strle F. Lyme borreliosis. Lancet. 2012;379:461–73. DOIPubMedGoogle Scholar
- Pritt BS, Mead PS, Johnson DK, Neitzel DF, Respicio-Kingry LB, Davis JP, Identification of a novel pathogenic Borrelia species causing Lyme borreliosis with unusually high spirochataemia: a descriptive study. Lancet Infect Dis. 2016;Feb 5:pii: S1473-3099(15)00464-8.
- Adams DA, Jajosky RA, Ajani U, Kriseman J, Sharp P, Onwen DH, Summary of notifiable diseases—United States, 2012. MMWR Morb Mortal Wkly Rep. 2014;61:1–121.PubMedGoogle Scholar
- Nelson CA, Saha S, Kugeler KJ, Delorey MJ, Shankar MB, Hinckley AF, Incidence of clinician-diagnosed Lyme disease, United States, 2005–2010. Emerg Infect Dis. 2015;21:1625–31. DOIPubMedGoogle Scholar
- Kugeler KJ, Farley GM, Forrester JD, Mead PS. Geographic distribution and expansion of human Lyme disease, United States. Emerg Infect Dis. 2015;21:1455–7. DOIPubMedGoogle Scholar
- Wormser GP, Dattwyler RJ, Shapiro ED, Halperin JJ, Steere AC, Klempner MS, The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2006;43:1089–134. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Recommendations for test performance and interpretation from the Second National Conference on serologic diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 1995;44:590–1.PubMedGoogle Scholar
- Seltzer EG, Shapiro ED. Misdiagnosis of Lyme disease: when not to order serologic tests. Pediatr Infect Dis J. 1996;15:762–3. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Notice to readers: caution regarding testing for Lyme disease. MMWR Morb Mortal Wkly Rep. 2005;54:125 [cited 2016 Mar 14]. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5405a6.htm
- Aguero-Rosenfeld ME, Wang G, Schwartz I, Wormser GP. Diagnosis of Lyme borreliosis. Clin Microbiol Rev. 2005;18:484–509. DOIPubMedGoogle Scholar
- Craven RB, Quan TJ, Bailey RE, Dattwyler R, Ryan RW, Sigal LH, Improved serodiagnostic testing for Lyme disease: results of a multicenter serologic evaluation. Emerg Infect Dis. 1996;2:136–40. DOIPubMedGoogle Scholar
- Branda JA, Linskey K, Kim YA, Steere AC, Ferraro MJ. Two-tiered antibody testing for Lyme disease with use of 2 enzyme immunoassays, a whole-cell sonicate enzyme immunoassay followed by a VlsE C6 peptide enzyme immunoassay. Clin Infect Dis. 2011;53:541–7. DOIPubMedGoogle Scholar
- Wormser GP, Schriefer M, Aguero-Rosenfeld ME, Levin A, Steere AC, Nadelman RB, Single-tier testing with the C6 peptide ELISA kit compared with two-tier testing for Lyme disease. Diagn Microbiol Infect Dis. 2013;75:9–15. DOIPubMedGoogle Scholar
- Molins CR, Sexton C, Young JW, Ashton LV, Pappert R, Beard CB, Collection and characterization of samples for establishment of a serum repository for Lyme disease diagnostic test development and evaluation. J Clin Microbiol. 2014;52:3755–62. DOIPubMedGoogle Scholar
- Wormser GP, Levin A, Soman S, Adenikinju O, Longo MV, Branda JA. Comparative cost-effectiveness of two-tiered testing strategies for serodiagnosis of Lyme disease with noncutaneous manifestations. J Clin Microbiol. 2013;51:4045–9. DOIPubMedGoogle Scholar
- Food and Drug Administration. Database: search on product code LSR [cited 2015 Jul 17]. http://www.accessdata.fda.gov/scripts/cdrh/devicesatfda/index.cfm
- Bacon RM, Biggerstaff BJ, Schriefer ME, Gilmore RD Jr, Philipp MT, Steere AC, Serodiagnosis of Lyme disease by kinetic enzyme-linked immunosorbent assay using recombinant VlsE1 or peptide antigens of Borrelia burgdorferi compared with 2-tiered testing using whole-cell lysates. J Infect Dis. 2003;187:1187–99. .DOIPubMedGoogle Scholar
- Liang FT, Steere AC, Marques AR, Johnson BJ, Miller JN, Philipp MT. Sensitive and specific serodiagnosis of Lyme disease by enzyme-linked immunosorbent assay with a peptide based on an immunodominant conserved region of Borrelia burgdorferi VlsE. J Clin Microbiol. 1999;37:3990–6.PubMedGoogle Scholar
- Aguero-Rosenfeld ME, Nowakowski J, Bittker S, Cooper D, Nadelman RB, Wormser GP. Evolution of the serologic response to Borrelia burgdorferi in treated patients with culture-confirmed erythema migrans. J Clin Microbiol. 1996;34:1–9.PubMedGoogle Scholar
- Nelson C, Elmendorf S, Mead P. Neoplasms misdiagnosed as “chronic Lyme disease.”. JAMA Intern Med. 2015;175:132–3. DOIPubMedGoogle Scholar
- Steere AC, Berardi VP, Weeks KE, Logigian EL, Ackermann R. Evaluation of the intrathecal antibody response to Borrelia burgdorferi as a diagnostic test for Lyme neuroborreliosis. J Infect Dis. 1990;161:1203–9. DOIPubMedGoogle Scholar
- Babady NE, Sloan LM, Vetter EA, Patel R, Binnicker MJ. Percent positive rate of Lyme real-time polymerase chain reaction in blood, cerebrospinal fluid, synovial fluid, and tissue. Diagn Microbiol Infect Dis. 2008;62:464–6. .DOIPubMedGoogle Scholar
- Molloy PJ, Persing DH, Berardi VP. False-positive results of PCR testing for Lyme disease. Clin Infect Dis. 2001;33:412–3. DOIPubMedGoogle Scholar
- Nocton JJ, Bloom BJ, Rutledge BJ, Persing DH, Logigian EL, Schmid CH, Detection of Borrelia burgdorferi DNA by polymerase chain reaction in cerebrospinal fluid in Lyme neuroborreliosis. J Infect Dis. 1996;174:623–7. DOIPubMedGoogle Scholar
- Molloy PJ, Telford SR III, Chowdri HR, Lepore TJ, Gugliotta JL, Weeks KE, Borrelia miyamotoi disease in the northeastern United States: a case series. Ann Intern Med. 2015;163:91–8. DOIPubMedGoogle Scholar
- Hilton E, DeVoti J, Benach JL, Halluska ML, White DJ, Paxton H, Seroprevalence and seroconversion for tick-borne diseases in a high-risk population in the northeast United States. Am J Med. 1999;106:404–9. DOIPubMedGoogle Scholar
- Kalish RA, McHugh G, Granquist J, Shea B, Ruthazer R, Steere AC. Persistence of immunoglobulin M or immunoglobulin G antibody responses to Borrelia burgdorferi 10–20 years after active Lyme disease. Clin Infect Dis. 2001;33:780–5. DOIPubMedGoogle Scholar
- Nadelman RB, Wormser GP. Reinfection in patients with Lyme disease. Clin Infect Dis. 2007;45:1032–8. DOIPubMedGoogle Scholar
- Lantos PM, Branda JA, Boggan JC, Chudgar SM, Wilson EA, Ruffin F, Poor positive predictive value of Lyme disease serologic testing in an area of low disease incidence. Clin Infect Dis. 2015;61:1374–80. DOIPubMedGoogle Scholar
- Masters EJ, Grigery CN, Masters RW. STARI, or Masters disease: Lone Star tick–vectored Lyme-like illness. Infect Dis Clin North Am. 2008;22:361–76. DOIPubMedGoogle Scholar
- Forrester JD, Brett M, Matthias J, Stanek D, Springs CB, Marsden-Haug N, Epidemiology of Lyme disease in low-incidence states. Ticks Tick Borne Dis. 2015;6:721–3. DOIPubMedGoogle Scholar
- Steere AC, Dhar A, Hernandez J, Fischer PA, Sikand VK, Schoen RT, Systemic symptoms without erythema migrans as the presenting picture of early Lyme disease. Am J Med. 2003;114:58–62. DOIPubMedGoogle Scholar
- Nelson C, Johnson B, Petersen J, Schriefer M, Beard CB, Petersen L, Concerns regarding a new culture method for Borrelia burgdorferi not approved for the diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 2014;63:333..PubMedGoogle Scholar
- Fallon BA, Pavlicova M, Coffino SW, Brenner C. A comparison of Lyme disease serologic test results from 4 laboratories in patients with persistent symptoms after antibiotic treatment. Clin Infect Dis. 2014;59:1705–10. DOIPubMedGoogle Scholar
- Johnson BJ, Pilgard MA, Russell TM. Assessment of new culture method for detection of Borrelia species from serum of Lyme disease patients. J Clin Microbiol. 2014;52:721–4. DOIPubMedGoogle Scholar
- Klempner MS, Schmid CH, Hu L, Steere AC, Johnson G, McCloud B, Intralaboratory reliability of serologic and urine testing for Lyme disease. Am J Med. 2001;110:217–9. DOIPubMedGoogle Scholar
- Marques A, Brown MR, Fleisher TA. Natural killer cell counts are not different between patients with post-Lyme disease syndrome and controls. Clin Vaccine Immunol. 2009;16:1249–50. DOIPubMedGoogle Scholar
- Molins CR, Ashton LV, Wormser GP, Hess AM, Delorey MJ, Mahapatra S, Development of a metabolic biosignature for detection of early Lyme disease. Clin Infect Dis. 2015;60:1767–75. DOIPubMedGoogle Scholar
- Schnell G, Boeuf A, Westermann B, Jaulhac B, Lipsker D, Carapito C, Discovery and targeted proteomics on cutaneous biopsies infected by Borrelia to investigate Lyme disease. Mol Cell Proteomics. 2015;14:1254–64. DOIPubMedGoogle Scholar
- Halpern MD, Molins CR, Schriefer M, Jewett MW. Simple objective detection of human Lyme disease infection using immuno-PCR and a single recombinant hybrid antigen. Clin Vaccine Immunol. 2014;21:1094–105. DOIPubMedGoogle Scholar
1These authors contributed equally to this article.
Page created: June 15, 2016
Page updated: November 30, 2016
Page reviewed: November 30, 2016
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.