Volume 22, Number 9—September 2016
Etymologia
Etymologia: β-Lactamase
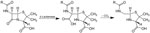
Enzymes that catalyze the cleavage of β-lactam rings in penicillins, cephalosporins, monobactams, and carbapenems were first described by Abraham and Chain in 1940. These enzymes confer resistance to β-lactam antibiotics on bacteria that produce them. β-lactamases (Figure) are ancient, theorized to have evolved 1–2 billion years ago, but the emergence and spread of penicillin-resistant staphylococci in hospitals in the 1950s showed how penicillin use could select producers from a population of nonproducers. “Lactam” is a portmanteau of “lactone” (from the Latin lactis, “milk,” since lactic acid was isolated from soured milk) and “amide.” The “β” refers to the nitrogen’s position on the second carbon in the ring. The suffix “-ase,” indicating an enzyme, is derived from “diastase” (from the Greek diastasis, “separation”), the first enzyme discovered in 1833 by Payen and Persoz.
References
- Abraham EP. A retrospective review of β-lactamases. J Chemother. 1991;3:67–74 .DOIPubMedGoogle Scholar
- Dixon M. The history of enzymes and of biological oxidations. In: Needham J, editor. The chemistry of life: lectures on the history of biochemistry. London: Cambridge University Press; 1970. p. 15–37.
- Dorland’s illustrated medical dictionary. 32nd ed. Philadelphia: Elsevier Saunders; 2012.
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 22, Number 9—September 2016
| EID Search Options |
|---|
|
|
|
|
|
|