Volume 25, Number 10—October 2019
Research
Comparison of Serologic Assays for Middle East Respiratory Syndrome Coronavirus
Figure 1
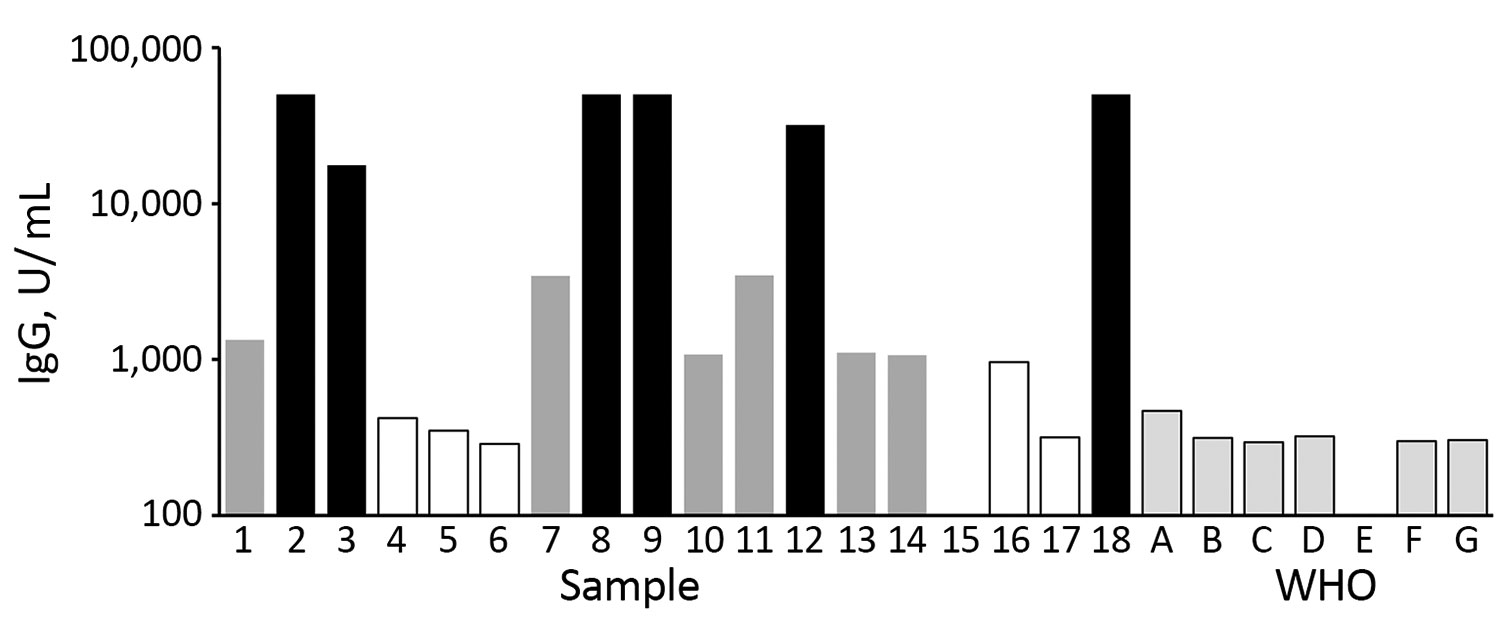
Figure 1. Pooling of serum samples based on their ELISA titers in study of serologic assays for Middle East respiratory syndrome coronavirus. Bar shading indicates the mean ELISA unit value of 2 independent experiments run in duplicate. Black bars represent samples used in pool A (high-positive); dark gray bars indicate samples used in pool B (medium-positive); white bars, and sample 15 with no visible bar, indicate samples used in pool C (low-positive). Pale gray bars with black outline indicate results from a set of negative control samples (WHO A–G). WHO, World Health Organization.
1Study participants who contributed data are listed at the end of this article.
Page created: September 17, 2019
Page updated: September 17, 2019
Page reviewed: September 17, 2019
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.