Tuberculosis among Children and Adolescents at HIV Treatment Centers in Sub-Saharan Africa
Anna M. Mandalakas
1
, Alexander W. Kay
1, Jason M. Bacha, Tara Devezin, Rachel Golin, Katherine R. Simon, Dilsher Dhillon, Sandile Dlamini, Andrew DiNardo, Mogo Matshaba, Jill Sanders, Lineo Thahane, Pauline M. Amuge, Saeed Ahmed, Moorine P. Sekadde, Neway G. Fida, Bhekumusa Lukhele, Nodumo Chidah, David Damba, Joseph Mhango, Moses Chodota, Makhorong Matsoso, Angelina Kayabu, Richard S. Wanless, and Gordon E. Schutze
Author affiliations: Texas Children’s Hospital, Houston, Texas, USA (A.M. Manadalakas, A.W. Kay, J.M. Bacha, T. Devezin, K.R. Simon, D. Dhillon, S. Dlamini, A. DiNardo, M. Matshaba, J. Sanders, L. Thahane, S. Ahmed, N. Chidah, D. Damba, M. Chodota, M. Matsoso, A. Kayabu, R.S. Wanless, G.E. Schutze); Baylor College of Medicine, Houston (A.M. Manadalakas, A.W. Kay, J.M. Bacha, T. Devezin, D. Dhillon, A. DiNardo, B. Lukhele, R.S. Wanless, G.E. Schutze); Baylor College of Medicine Children’s Foundation Swaziland, Mbabane, Swaziland (A.W. Kay, S. Dlamini, B. Lukhele); Baylor College of Medicine Children's Foundation Tanzania, Mbeya, Tanzania (J.M. Bacha); US Agency for International Development, Washington, DC, USA (R. Golin); Baylor College of Medicine Children’s Foundation Malawi, Lilongwe, Malawi (K.R. Simon, S. Ahmed, J. Mhango); Technical Support to PEPFAR Programs in the Southern Africa Region, Lilongwe (K.R. Simon, S. Ahmed, J. Mhango); Botswana-Baylor Children’s Clinical Centre of Excellence, Gaborone, Botswana (M. Matshaba, N. Chidah); Baylor College of Medicine Children’s Foundation Lesotho, Maseru, Lesotho (J. Sanders, L. Thahane, M. Matsoso); Baylor College of Medicine Children’s Foundation Uganda, Kampala, Uganda (P.M. Amuge, D. Damba); National Tuberculosis and Leprosy Programme, Kampala, Uganda (M.P. Sekkade); US Agency for International Development, Pretoria, South Africa (N.G. Fida); Baylor College of Medicine Children’s Foundation Tanzania, Mwanza, Tanzania (M. Chodota, A. Kayabu)
Main Article
Figure 4
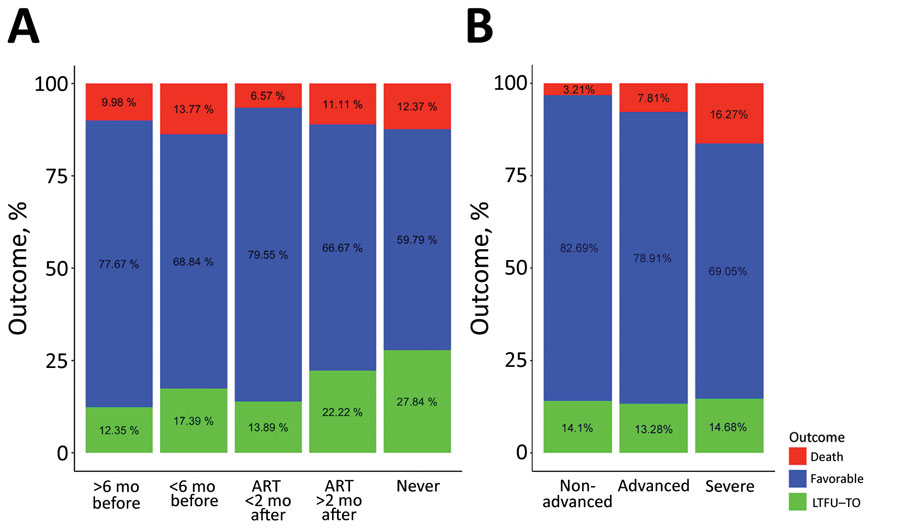
Figure 4. Bivariate analyses of factors relating to TB treatment outcome in HIV-infected children and adolescents, 2013–2017. A) ART treatment category: received ART >6 months or <6 months before TB diagnosis or started on ART <2 months or >2 months after TB diagnosis. B) Immune status. Advanced immunosuppression was define as a CD4 percentage of <25% in children <5 years of age or CD4 count <200 cells/mm3 in children >5 years of age. ART, antiretroviral therapy; TB, tuberculosis.
Main Article
Page created: September 10, 2020
Page updated: December 01, 2020
Page reviewed: December 01, 2020
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.
